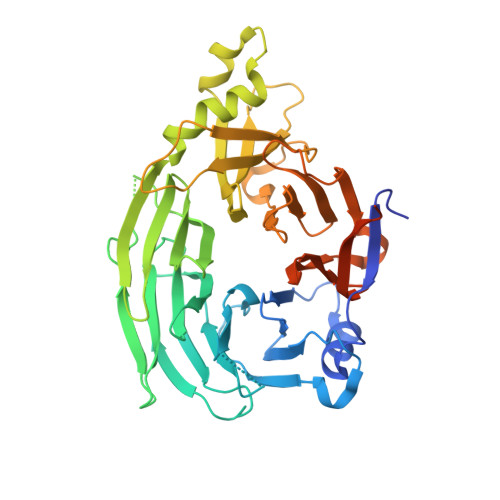
Structural and functional analysis of Nup133 domains reveals modular building blocks of the nuclear pore complex.
Berke, I.C., Boehmer, T., Blobel, G., Schwartz, T.U.(2004) J Cell Biol 167: 591-597
- PubMed: 15557116 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1083/jcb.200408109
- Primary Citation Related Structures:
1XKS - PubMed Abstract:
Nucleocytoplasmic transport occurs through nuclear pore complexes (NPCs) whose complex architecture is generated from a set of only approximately 30 proteins, termed nucleoporins. Here, we explore the domain structure of Nup133, a nucleoporin in a conserved NPC subcomplex that is crucial for NPC biogenesis and is believed to form part of the NPC scaffold. We show that human Nup133 contains two domains: a COOH-terminal domain responsible for its interaction with its subcomplex through Nup107; and an NH2-terminal domain whose crystal structure reveals a seven-bladed beta-propeller. The surface properties and conservation of the Nup133 beta-propeller suggest it may mediate multiple interactions with other proteins. Other beta-propellers are predicted in a third of all nucleoporins. These and several other repeat-based motifs appear to be major elements of nucleoporins, indicating a level of structural repetition that may conceptually simplify the assembly and disassembly of this huge protein complex.
- Laboratory of Cell Biology, Howard Hughes Medical Institute, The Rockefeller University, New York, NY 10021, USA.
Organizational Affiliation: