Crystal structure of the uncharacterized protein ST1454 from Sulfolobus tokodaii.
Sasaki, T., Tanaka, Y., Yasutake, Y., Yao, M., Tanaka, I., Tsumoto, K., Kumagai, I.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 177aa long conserved hypothetical protein (ST1454) | 177 | Sulfurisphaera tokodaii str. 7 | Mutation(s): 4 Gene Names: ST1454 EC: 2.5.1.17 | 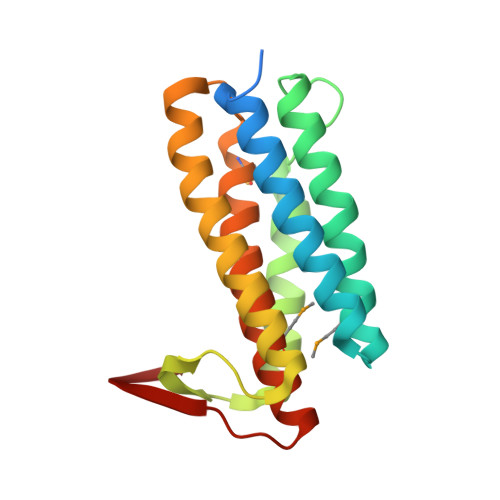 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q970Z7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| POG Download:Ideal Coordinates CCD File | B [auth A] | (20S)-2,5,8,11,14,17-HEXAMETHYL-3,6,9,12,15,18-HEXAOXAHENICOSANE-1,20-DIOL C21 H44 O8 OWRNLGZKEZSHGO-QAWAPDBNSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 84.67 | α = 90 |
| b = 84.67 | β = 90 |
| c = 84.67 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| SOLVE | phasing |
| CNS | refinement |