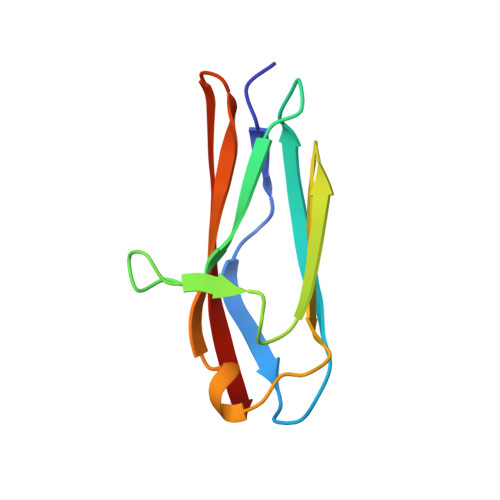
Structure and stability of an immunoglobulin superfamily domain from twitchin, a muscle protein of the nematode Caenorhabditis elegans.
Fong, S., Hamill, S.J., Proctor, M., Freund, S.M., Benian, G.M., Chothia, C., Bycroft, M., Clarke, J.(1996) J Mol Biology 264: 624-639
- PubMed: 8969309 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1996.0665
- Primary Citation Related Structures:
1WIT, 1WIU - PubMed Abstract:
The NMR solution structure of an immunoglobulin superfamily module of twitchin (Ig 18') has been determined and the kinetic and equilibrium folding behaviour characterised. Thirty molecular coordinates were calculated using a hybrid distance geometry-simulated annealing protocol based on 1207 distance and 48 dihedral restraints. The atomic rms distributions about the mean coordinate for the ensemble of structures is 0.55( +/- 0.09) A for backbone atoms and 1.10( +/- 0.08) A for all heavy atoms. The protein has a topology very similar to that of telokin and the titin Ig domains and thus it falls into the I set of the immunoglobulin superfamily. The close agreement between the predicted and observed structures of Ig 18' demonstrates clearly that the I set profile can be applied in the structure prediction of immunoglobulin-like domains of diverse modular proteins. Folding studies reveal that the protein has relatively low thermodynamic stability, deltaG(H2O)U-F = 4.0 kcal mol(-1) at physiological pH. Unfolding studies suggest that the protein has considerable kinetic stability, the half life of the unfolding is greater than 40 minutes in the absence of denaturant.
- Centre for Protein Engineering, MRC Unit of Protein Folding and Design, Cambridge, UK.
Organizational Affiliation: