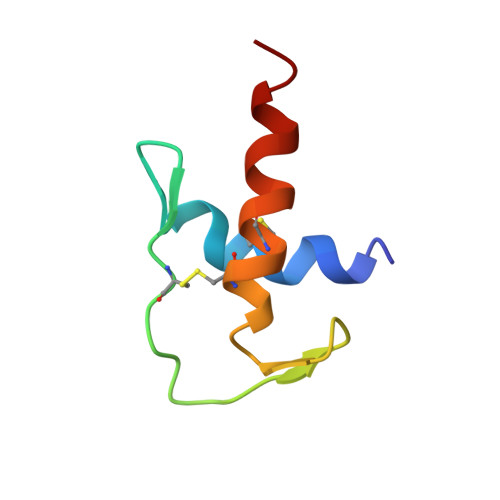
The Solution Structure of a Chimeric Lekti Domain Reveals a Chameleon Sequence
Tidow, H., Lauber, T., Vitzithum, K., Sommerhoff, C., Roesch, P., Marx, U.C.(2004) Biochemistry 43: 11238
- PubMed: 15366933 Search on PubMed
- DOI: https://doi.org/10.1021/bi0492399
- Primary Citation Related Structures:
1UUC, 1UVF, 1UVG - PubMed Abstract:
The conversion of an alpha-helical to a beta-strand conformation and the presence of chameleon sequences are fascinating from the perspective that such structural features are implicated in the induction of amyloid-related fatal diseases. In this study, we have determined the solution structure of a chimeric domain (Dom1PI) from the multidomain Kazal-type serine proteinase inhibitor LEKTI using multidimensional NMR spectroscopy. This chimeric protein was constructed to investigate the reasons for differences in the folds of the homologous LEKTI domains 1 and 6 [Lauber, T., et al. (2003) J. Mol. Biol. 328, 205-219]. In Dom1PI, two adjacent phenylalanine residues (F28 and F29) of domain 1 were substituted with proline and isoleucine, respectively, as found in the corresponding P4' and P5' positions of domain 6. The three-dimensional structure of Dom1PI is significantly different from the structure of domain 1 and closely resembles the structure of domain 6, despite the sequence being identical to that of domain 1 except for the two substituted phenylalanine residues and being only 31% identical to the sequence of domain 6. The mutation converted a short 3(10)-helix into an extended loop conformation and parts of the long COOH-terminal alpha-helix of domain 1 into a beta-hairpin structure. The latter conformational change occurs in a sequence stretch distinct from the region containing the substituted residues. Therefore, this switch from an alpha-helical structure to a beta-hairpin structure indicates a chameleon sequence of seven residues. We conclude that the secondary structure of Dom1PI is determined not only by the local protein sequence but also by nonlocal interactions.
- Lehrstuhl für Biopolymere, Universität Bayreuth, Universitätsstrasse 30, 95447 Bayreuth, Germany.
Organizational Affiliation: