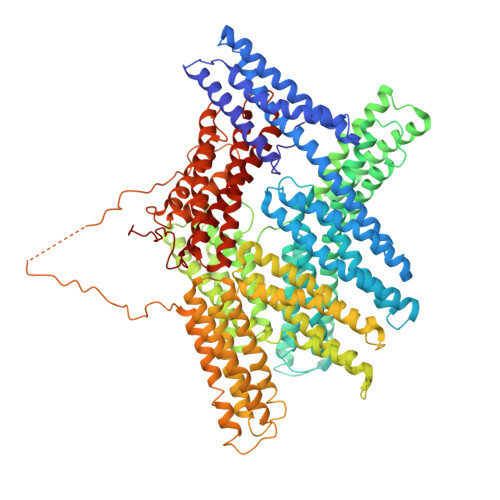
Structural basis for vinculin activation at sites of cell adhesion.
Bakolitsa, C., Cohen, D.M., Bankston, L.A., Bobkov, A.A., Cadwell, G.W., Jennings, L., Critchley, D.R., Craig, S.W., Liddington, R.C.(2004) Nature 430: 583-586
- PubMed: 15195105 Search on PubMed
- DOI: https://doi.org/10.1038/nature02610
- Primary Citation Related Structures:
1ST6 - PubMed Abstract:
Vinculin is a highly conserved intracellular protein with a crucial role in the maintenance and regulation of cell adhesion and migration. In the cytosol, vinculin adopts a default autoinhibited conformation. On recruitment to cell-cell and cell-matrix adherens-type junctions, vinculin becomes activated and mediates various protein-protein interactions that regulate the links between F-actin and the cadherin and integrin families of cell-adhesion molecules. Here we describe the crystal structure of the full-length vinculin molecule (1,066 amino acids), which shows a five-domain autoinhibited conformation in which the carboxy-terminal tail domain is held pincer-like by the vinculin head, and ligand binding is regulated both sterically and allosterically. We show that conformational changes in the head, tail and proline-rich domains are linked structurally and thermodynamically, and propose a combinatorial pathway to activation that ensures that vinculin is activated only at sites of cell adhesion when two or more of its binding partners are brought into apposition.
- Program on Cell Adhesion, The Burnham Institute, 10901 North Torrey Pines Road, La Jolla, California 92037, USA.
Organizational Affiliation: