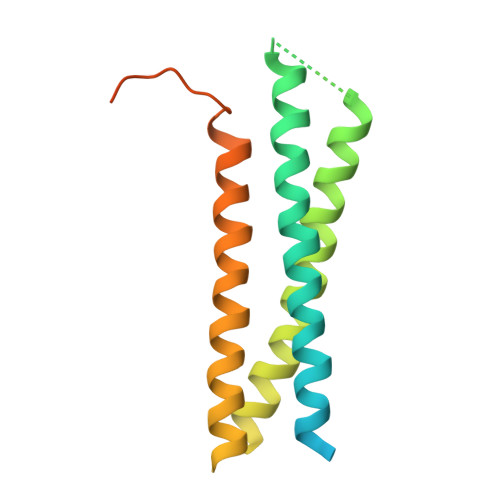
Crystal structure of the Habc domain of neuronal syntaxin from the squid Loligo pealei reveals conformational plasticity at its C-terminus
Bracher, A., Weissenhorn, W.(2004) BMC Struct Biol 4: 6-6
- PubMed: 15113421 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1186/1472-6807-4-6
- Primary Citation Related Structures:
1S94 - PubMed Abstract:
Intracellular membrane fusion processes are mediated by the spatial and temporal control of SNARE complex assembly that results in the formation of a four-helical bundle, composed of one vesicle SNARE and three target membrane SNARE polypeptide chains. Syntaxins are essential t-SNAREs and are characterized by an N-terminal Habc domain, a flexible linker region, a coiled-coil or SNARE motif and a membrane anchor. The N-terminal Habc domain fulfills important regulatory functions while the coiled-coil motif, present in all SNAREs, is sufficient for SNARE complex formation, which is thought to drive membrane fusion. Here we report the crystal structure of the Habc domain of neuronal syntaxin from the squid Loligo pealei, s-syntaxin. Squid Habc crystallizes as a dimer and the monomer structure consists of a three-helical bundle. One molecule is strikingly similar to mammalian syntaxin 1A while the second one shows a structural deviation from the common fold in that the C-terminal part of helix C unwinds and adopts an extended conformation. Conservation of surface residues indicates that the cytosolic part of s-syntaxin can adopt an auto-inhibitory closed conformation that may bind squid neuronal Sec1, s-Sec1, in the same manner as observed in structure of the rat nSec1/syntaxin 1A complex. Furthermore, despite the overall structural similarity, the observed changes at the C-terminus of one molecule indicate structural plasticity in neuronal syntaxin. Implications of the structural conservation and the changes are discussed with respect to potential Habc domain binding partners such as Munc13, which facilitates the transition from the closed to the open conformation.
- European Molecular Biology Laboratory, 6 rue Jules Horowitz, 38042 Grenoble, France. bracher@biochem.mpg.de
Organizational Affiliation: