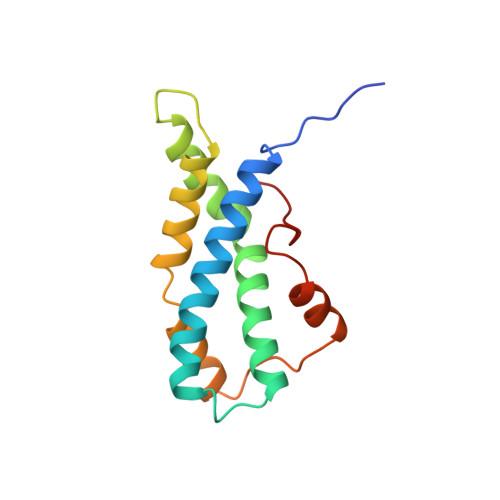
The crystal structure of lysin, a fertilization protein.
Shaw, A., McRee, D.E., Vacquier, V.D., Stout, C.D.(1993) Science 262: 1864-1867
- PubMed: 8266073 Search on PubMed
- DOI: https://doi.org/10.1126/science.8266073
- Primary Citation Related Structures:
1LIS - PubMed Abstract:
Lysin, a protein from abalone sperm, creates a hole in the envelope of the egg, permitting the sperm to pass through the envelope and fuse with the egg. The structure of lysin, refined at 1.9 angstroms resolution, reveals an alpha-helical, amphipathic molecule. The surface of the protein exhibits three features: two tracks of basic residues that span the length of the molecule, a solvent-exposed cluster of aromatic and aliphatic amino acids, and an extended amino-terminal hypervariable domain that is species-specific. The structure suggests possible mechanisms of action.
- Department of Molecular Biology, Scripps Research Institute, La Jolla, CA 92037-1093.
Organizational Affiliation: