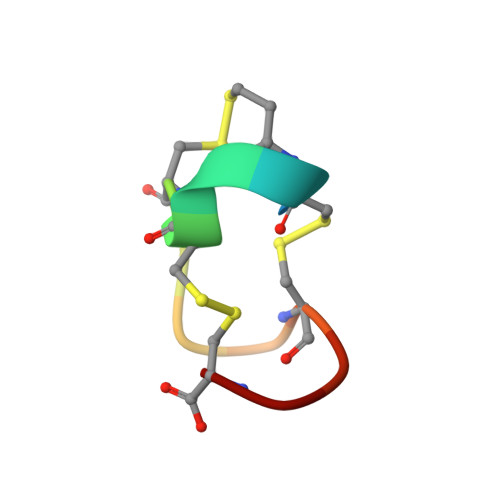
Molecular structure of the toxin domain of heat-stable enterotoxin produced by a pathogenic strain of Escherichia coli. A putative binding site for a binding protein on rat intestinal epithelial cell membranes.
Ozaki, H., Sato, T., Kubota, H., Hata, Y., Katsube, Y., Shimonishi, Y.(1991) J Biological Chem 266: 5934-5941
- PubMed: 2005130 Search on PubMed
- Primary Citation Related Structures:
1ETN - PubMed Abstract:
Heat-stable enterotoxins are a family of toxin peptides that are produced by enterotoxigenic Escherichia coli and consist of 18 and 19 amino acid residues (Aimoto, S., Takao, T., Shimonishi, Y., Hara, S., Takeda, T., Takeda, Y., and Miwatani, T. (1982) Eur. J. Biochem. 129, 257-263). A synthetic fully toxic analog of the enterotoxin, Mpr5-STp(5-17), where Mpr is beta-mercaptopropionic acid and which consists of 13 amino acid residues from Cys5 to Cys17 in a heat-stable enterotoxin but is deaminated at its N terminus (Kubota, H., Hidaka, Y., Ozaki, H., Ito, H., Hirayama, T., Takeda, Y., and Shimonishi, Y. (1989) Biochem. Biophys. Res. Commun. 161, 229-235), has been crystalized from water, and its crystal structure has been solved by a direct method and refined by least square procedures to give an R factor of 0.089. The crystal belongs to the orthorhombic space group P2(1)2(1)2(1) with unit cell constants a = 21.010 (2) A, b = 27.621 (4) A, and c = 12.781 (1) A. The asymmetric unit of the crystals contains one peptide molecule with 13 water molecules. A right-hand spiral peptide backbone extends throughout the molecule. Three beta-turns are located along this spiral and fixed tightly by three intramolecular disulfide linkages. The actual structure predicts the biniding region on the enterotoxin to the receptor protein on the membrane of rat intestinal epithelial cells.
- Institute for Protein Research, Osaka University, Japan.
Organizational Affiliation: