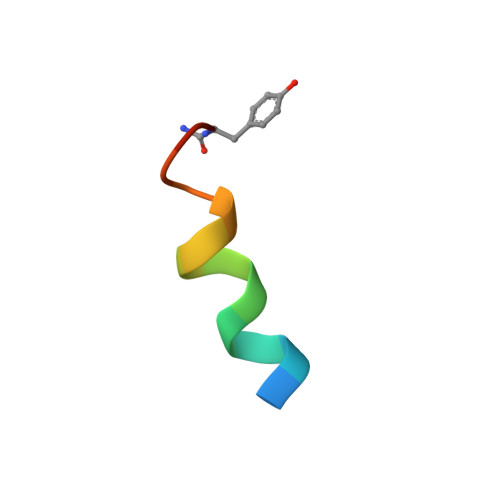
Stabilization of the helical structure of Y2-selective analogues of neuropeptide Y by lactam bridges.
Yao, S., Smith-White, M.A., Potter, E.K., Norton, R.S.(2002) J Med Chem 45: 2310-2318
- PubMed: 12014969 Search on PubMed
- DOI: https://doi.org/10.1021/jm010543z
- Primary Citation Related Structures:
1D0W, 1D1E, 1D1F - PubMed Abstract:
The importance of helical structure in an analogue of NPY selective for the Y2 receptor, Ac[Leu28,31]NPY24-36, has been investigated by introducing a lactam bridge between positions 28 and 32. The resulting analogue, Ac-cyclo28/32[Ala24,Lys28,Leu31,Glu32]NPY24-36, is a potent Y2-selective agonist. Structural analysis by NMR shows that this analogue forms a helical structure in a 40% trifluoroethanol/water mixture, whereas in water only the region around the lactam bridge (Lys28-Glu32) adopts helical-like structure, with both N- and C-termini being poorly defined. The observation of well-defined helical structure in aqueous TFE contrasts with that reported for a similar analogue, Ac-cyclo28/32[Lys28,Glu32]NPY25-36 (Rist et al. FEBS Lett. 1996, 394, 169-173), which consisted of a hairpin-like structure that brought the N- and C-termini into proximity. We have therefore determined the structures of this analogue, as well as those of Ac-cyclo28/32[Ala24,Lys28,Leu31,Glu32]NPY24-36 and Ac-cyclo28/32[Ala24,Lys28,Glu32]NPY24-36, under identical solution conditions (30% TFE/H2O mixture at 308 K) and find essentially the same helical structure in all three peptides. These findings support the proposal that these Y2-selective analogues adopt a helical structure when bound to the Y2 receptor.
- Biomolecular Research Institute, 343 Royal Parade, Parkville, Victoria 3052, Australia.
Organizational Affiliation: