Cobalamin decyanation by the membrane transporter BtuM.
Martinez Felices, J.M., Barreto, Y.B., Thangaratnarajah, C., Whittaker, J.J., Alencar, A.M., Guskov, A., Slotboom, D.J.(2024) Structure 32: 1165-1173.e3
- PubMed: 38733996
- DOI: https://doi.org/10.1016/j.str.2024.04.014
- Primary Citation of Related Structures:
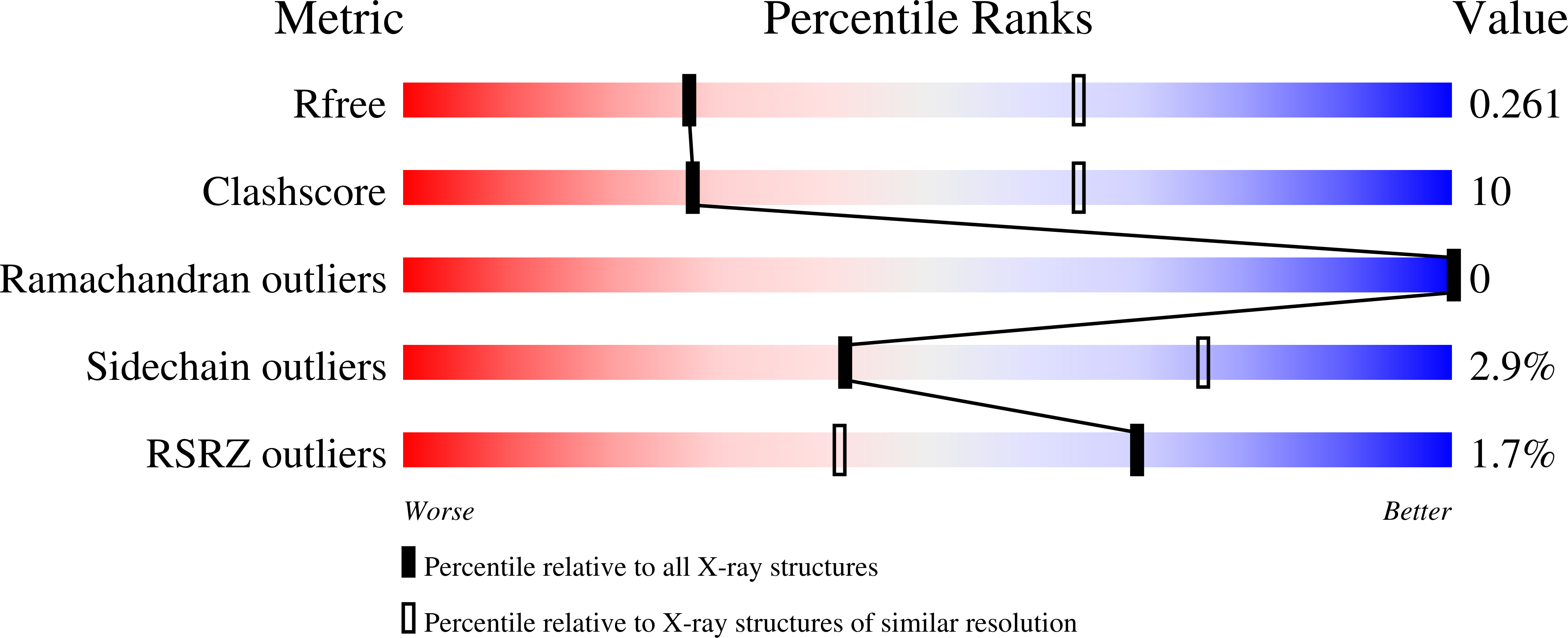
8C7P - PubMed Abstract:
BtuM is a bacterial cobalamin transporter that binds the transported substrate in the base-off state, with a cysteine residue providing the α-axial coordination of the central cobalt ion via a sulfur-cobalt bond. Binding leads to decyanation of cobalamin variants with a cyano group as the β-axial ligand. Here, we report the crystal structures of untagged BtuM bound to two variants of cobalamin, hydroxycobalamin and cyanocobalamin, and unveil the native residue responsible for the β-axial coordination, His28. This coordination had previously been obscured by non-native histidines of His-tagged BtuM. A model in which BtuM initially binds cobinamide reversibly with low affinity (K D = 4.0 μM), followed by the formation of a covalent bond (rate constant of 0.163 s -1 ), fits the kinetics data of substrate binding and decyanation of the cobalamin precursor cobinamide by BtuM. The covalent binding mode suggests a mechanism not used by any other transport protein.
Organizational Affiliation:
Groningen Biomolecular and Biotechnology Institute (GBB), University of Groningen, Nijenborgh 4, Groningen 9474 AG, the Netherlands.