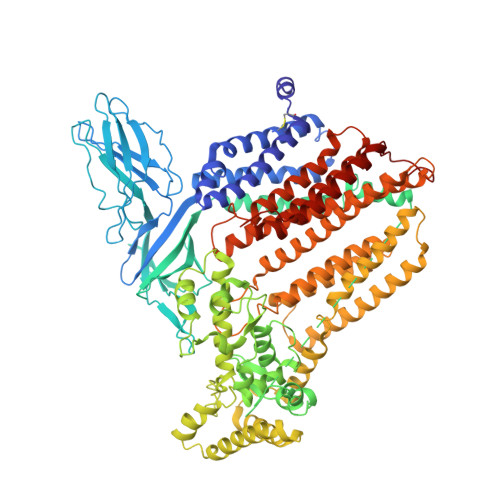
Cryo-EM of CcsBA reveals the basis for cytochrome c biogenesis and heme transport.
Mendez, D.L., Lowder, E.P., Tillman, D.E., Sutherland, M.C., Collier, A.L., Rau, M.J., Fitzpatrick, J.A.J., Kranz, R.G.(2022) Nat Chem Biol 18: 101-108
- PubMed: 34931065
- DOI: https://doi.org/10.1038/s41589-021-00935-y
- Primary Citation of Related Structures:
7S9Y, 7S9Z - PubMed Abstract:
Although the individual structures and respiratory functions of cytochromes are well studied, the structural basis for their assembly, including transport of heme for attachment, are unknown. We describe cryo-electron microscopy (cryo-EM) structures of CcsBA, a bifunctional heme transporter and cytochrome c (cyt c) synthase. Models built from the cryo-EM densities show that CcsBA is trapped with heme in two conformations, herein termed the closed and open states. The closed state has heme located solely at a transmembrane (TM) site, with a large periplasmic domain oriented such that access of heme to the cytochrome acceptor is denied. The open conformation contains two heme moieties, one in the TM-heme site and another in an external site (P-heme site). The presence of heme in the periplasmic site at the base of a chamber induces a large conformational shift that exposes the heme for reaction with apocytochrome c (apocyt c). Consistent with these structures, in vivo and in vitro cyt c synthase studies suggest a mechanism for transfer of the periplasmic heme to cytochrome.
Organizational Affiliation:
Department of Biology, Washington University in St. Louis, St. Louis, MO, USA.