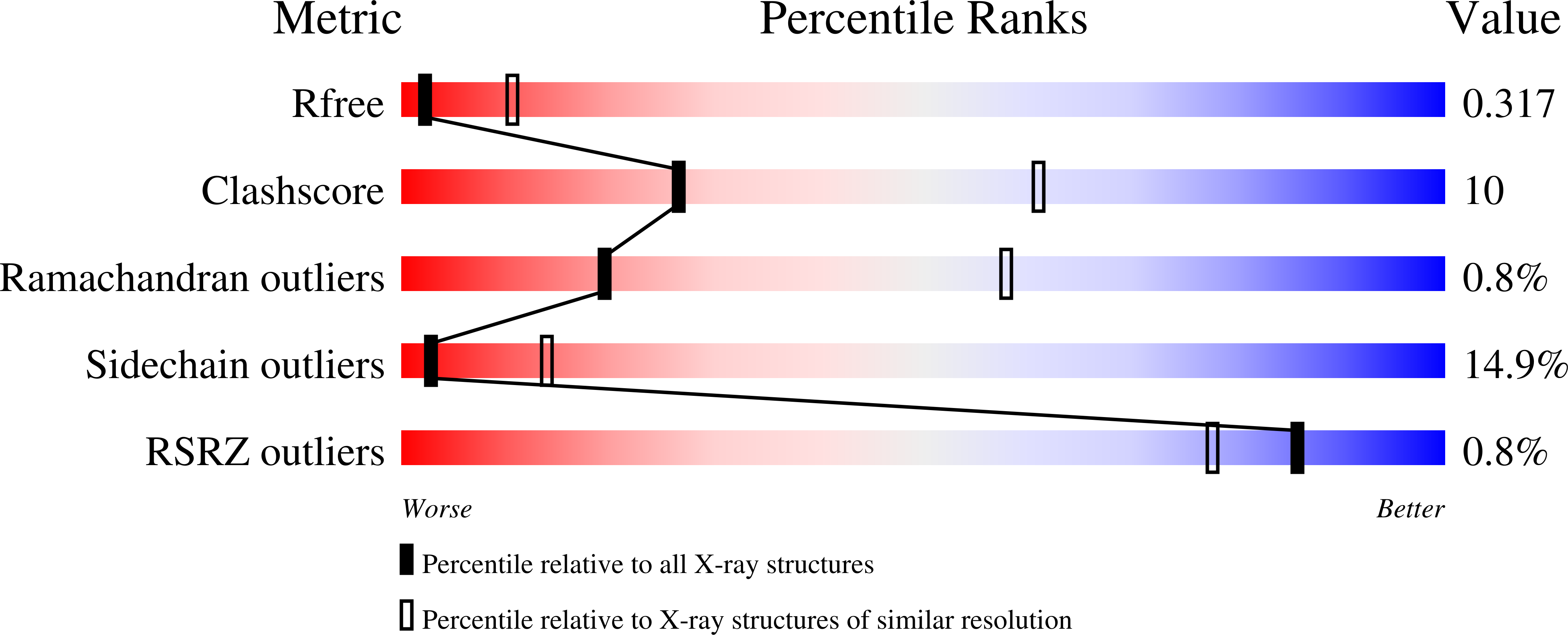
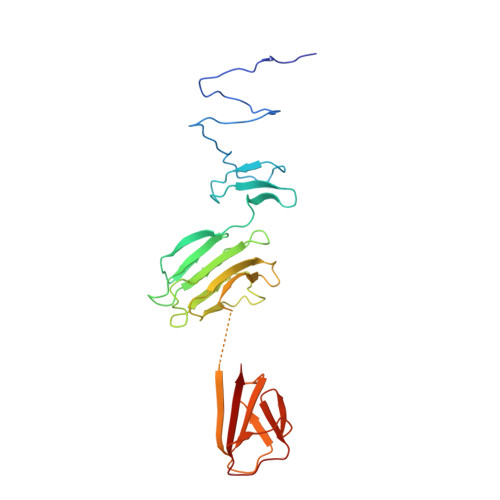
Structure of Escherichia coli O157:H7 bacteriophage CBA120 tailspike protein 4 baseplate anchor and tailspike assembly domains (TSP4-N).
Chao, K.L., Shang, X., Greenfield, J., Linden, S.B., Alreja, A.B., Nelson, D.C., Herzberg, O.(2022) Sci Rep 12: 2061-2061
- PubMed: 35136138
- DOI: https://doi.org/10.1038/s41598-022-06073-2
- Primary Citation of Related Structures:
7REJ, 7RFO, 7RFV - PubMed Abstract:
Four tailspike proteins (TSP1-4) of Escherichia coli O157:H7 bacteriophage CBA120 enable infection of multiple hosts. They form a branched complex that attaches to the tail baseplate. Each TSP recognizes a different lipopolysaccharide on the membrane of a different bacterial host. The 335 N-terminal residues of TSP4 promote the assembly of the TSP complex and anchor it to the tail baseplate. The crystal structure of TSP4-N 335 reveals a trimeric protein comprising four domains. The baseplate anchor domain (AD) contains an intertwined triple-stranded β-helix. The ensuing XD1, XD2 and XD3 β-sheet containing domains mediate the binding of TSP1-3 to TSP4. Each of the XD domains adopts the same fold as the respective XD domains of bacteriophage T4 gp10 baseplate protein, known to engage in protein-protein interactions via its XD2 and XD3 domains. The structural similarity suggests that XD2 and XD3 of TSP4 also function in protein-protein interactions. Analytical ultracentrifugation analyses of TSP4-N 335 and of domain deletion proteins showed how TSP4-N 335 promotes the formation of the TSP quaternary complex. TSP1 and TSP2 bind directly to TSP4 whereas TSP3 binding requires a pre-formed TSP4-N 335 :TSP2 complex. A 3-dimensional model of the bacteriophage CBA120 TSP complex has been developed based on the structural and ultracentrifuge information.
Organizational Affiliation:
Institute for Bioscience and Biotechnology Research, University of Maryland, 9600 Gudelsky Drive, Rockville, MD, 20850, USA.