Structure of the small Dictyostelium discoideum myosin light chain MlcB provides insights into MyoB IQ motif recognition.
Liburd, J., Chitayat, S., Crawley, S.W., Munro, K., Miller, E., Denis, C.M., Spencer, H.L., Cote, G.P., Smith, S.P.(2014) J Biol Chem 289: 17030-17042
- PubMed: 24790102
- DOI: https://doi.org/10.1074/jbc.M113.536532
- Primary Citation of Related Structures:
2M1U - PubMed Abstract:
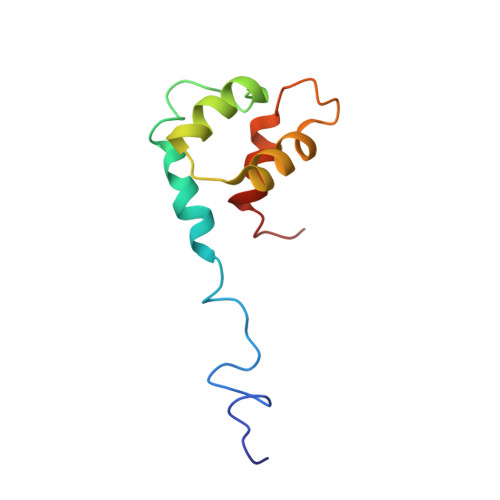
Dictyostelium discoideum MyoB is a class I myosin involved in the formation and retraction of membrane projections, cortical tension generation, membrane recycling, and phagosome maturation. The MyoB-specific, single-lobe EF-hand light chain MlcB binds the sole IQ motif of MyoB with submicromolar affinity in the absence and presence of Ca(2+). However, the structural features of this novel myosin light chain and its interaction with its cognate IQ motif remain uncharacterized. Here, we describe the NMR-derived solution structure of apoMlcB, which displays a globular four-helix bundle. Helix 1 adopts a unique orientation when compared with the apo states of the EF-hand calcium-binding proteins calmodulin, S100B, and calbindin D9k. NMR-based chemical shift perturbation mapping identified a hydrophobic MyoB IQ binding surface that involves amino acid residues in helices I and IV and the functional N-terminal Ca(2+) binding loop, a site that appears to be maintained when MlcB adopts the holo state. Complementary mutagenesis and binding studies indicated that residues Ile-701, Phe-705, and Trp-708 of the MyoB IQ motif are critical for recognition of MlcB, which together allowed the generation of a structural model of the apoMlcB-MyoB IQ complex. We conclude that the mode of IQ motif recognition by the novel single-lobe MlcB differs considerably from that of stereotypical bilobal light chains such as calmodulin.
Organizational Affiliation:
From the Department of Biomedical and Molecular Sciences and.