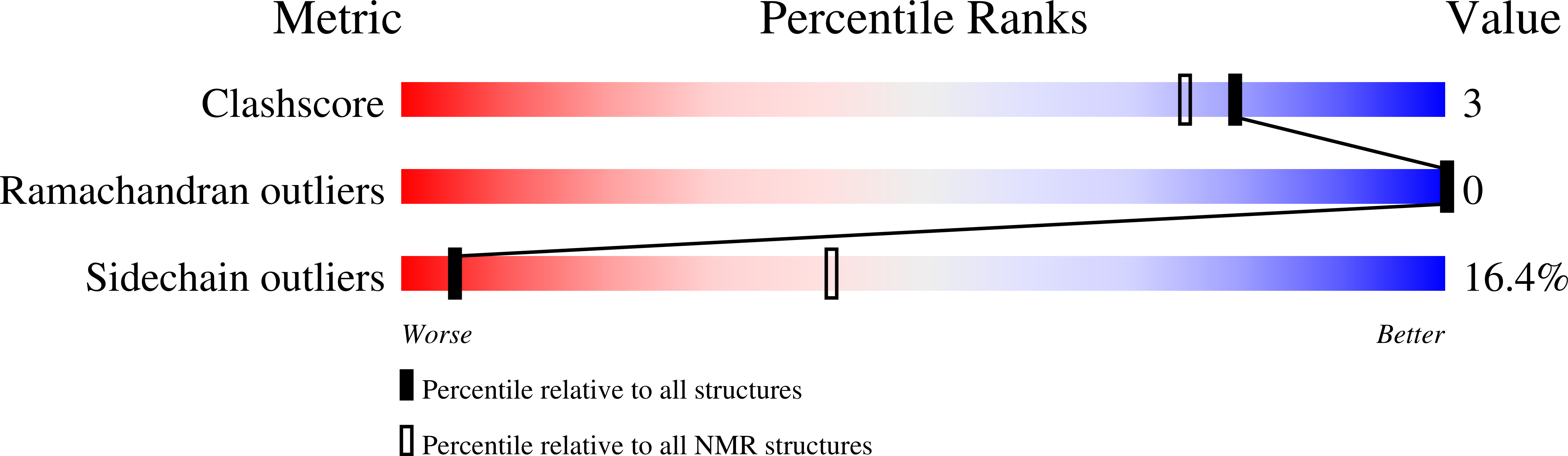Biophysical Insight on the Membrane Insertion of an Arginine-Rich Cell-Penetrating Peptide.
Jobin, M.L., Vamparys, L., Deniau, R., Grelard, A., Mackereth, C.D., Fuchs, P.F.J., Alves, I.D.(2019) Int J Mol Sci 20
- PubMed: 31505894
- DOI: https://doi.org/10.3390/ijms20184441
- Primary Citation of Related Structures:
6RQS - PubMed Abstract:
Cell-penetrating peptides (CPPs) are short peptides that can translocate and transport cargoes into the intracellular milieu by crossing biological membranes. The mode of interaction and internalization of cell-penetrating peptides has long been controversial. While their interaction with anionic membranes is quite well understood, the insertion and behavior of CPPs in zwitterionic membranes, a major lipid component of eukaryotic cell membranes, is poorly studied. Herein, we investigated the membrane insertion of RW16 into zwitterionic membranes, a versatile CPP that also presents antibacterial and antitumor activities. Using complementary approaches, including NMR spectroscopy, fluorescence spectroscopy, circular dichroism, and molecular dynamic simulations, we determined the high-resolution structure of RW16 and measured its membrane insertion and orientation properties into zwitterionic membranes. Altogether, these results contribute to explaining the versatile properties of this peptide toward zwitterionic lipids.
Organizational Affiliation:
Institute of Chemistry & Biology of Membranes & Nanoobjects (CBMN), CNRS UMR5248, University of Bordeaux, Bordeaux INP, allée Geoffroy St-Hilaire, 33600 Pessac, France. marie-lise.jobin@u-bordeaux.fr.