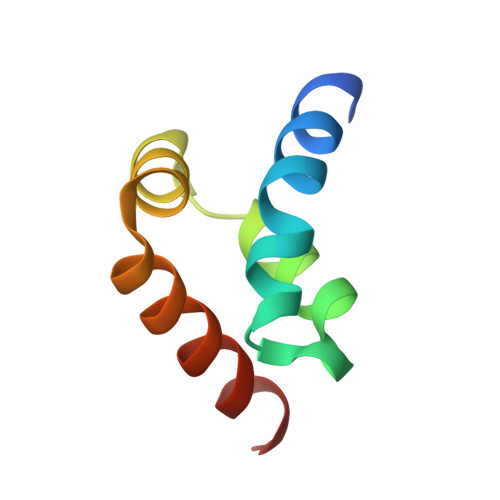
The MLLE domain of the ubiquitin ligase UBR5 binds to its catalytic domain to regulate substrate binding.
Munoz-Escobar, J., Matta-Camacho, E., Kozlov, G., Gehring, K.(2015) J Biol Chem 290: 22841-22850
- PubMed: 26224628
- DOI: https://doi.org/10.1074/jbc.M115.672246
- Primary Citation of Related Structures:
3NTW - PubMed Abstract:
E3 ubiquitin ligases catalyze the transfer of ubiquitin from an E2-conjugating enzyme to a substrate. UBR5, homologous to the E6AP C terminus (HECT)-type E3 ligase, mediates the ubiquitination of proteins involved in translation regulation, DNA damage response, and gluconeogenesis. In addition, UBR5 functions in a ligase-independent manner by prompting protein/protein interactions without ubiquitination of the binding partner. Despite recent functional studies, the mechanisms involved in substrate recognition and selective ubiquitination of its binding partners remain elusive. The C terminus of UBR5 harbors the HECT catalytic domain and an adjacent MLLE domain. MLLE domains mediate protein/protein interactions through the binding of a conserved peptide motif, termed PAM2. Here, we characterize the binding properties of the UBR5 MLLE domain to PAM2 peptides from Paip1 and GW182. The crystal structure with a Paip1 PAM2 peptide reveals the network of hydrophobic and ionic interactions that drive binding. In addition, we identify a novel interaction of the MLLE domain with the adjacent HECT domain mediated by a PAM2-like sequence. Our results confirm the role of the MLLE domain of UBR5 in substrate recruitment and suggest a potential role in regulating UBR5 ligase activity.
Organizational Affiliation:
From the Department of Biochemistry, Groupe de Recherche Axé sur la Structure des Protéines, McGill University, Montréal, Québec H3G 0B1, Canada.