Diselenide crosslinks for enhanced and simplified oxidative protein folding
Mousa, R., Hidmi, T., Pomyalov, S., Lansky, S., Khouri, L., Shalev, D.E., Shoham, G., Metanis, N.(2021) Commun Chem 4: 30
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
(2021) Commun Chem 4: 30
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Prothrombin | A [auth HHH] | 259 | Bos taurus | Mutation(s): 0 EC: 3.4.21.5 | 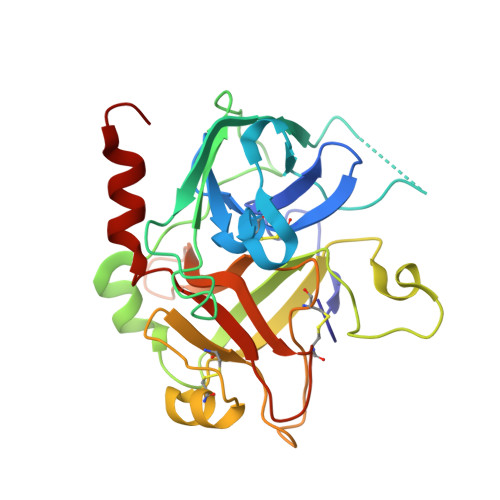 |
UniProt | |||||
Find proteins for P00735 (Bos taurus) Explore P00735 Go to UniProtKB: P00735 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00735 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Hirudin variant-1 | B [auth III] | 65 | Hirudo medicinalis | Mutation(s): 2 | 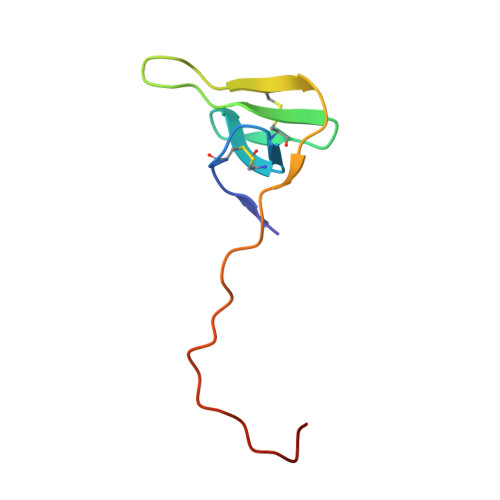 |
UniProt | |||||
Find proteins for P01050 (Hirudo medicinalis) Explore P01050 Go to UniProtKB: P01050 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P01050 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Prothrombin | C [auth LLL] | 49 | Bos taurus | Mutation(s): 0 EC: 3.4.21.5 | 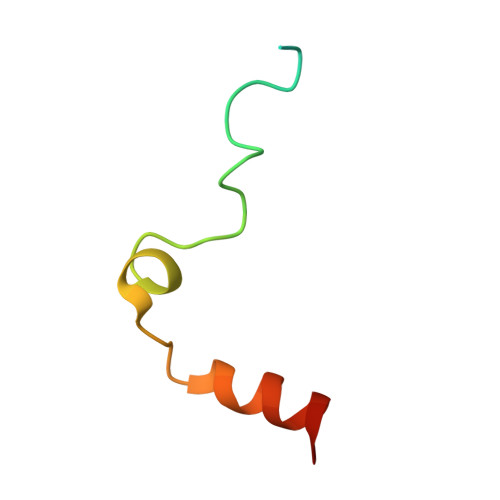 |
UniProt | |||||
Find proteins for P00735 (Bos taurus) Explore P00735 Go to UniProtKB: P00735 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00735 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NA Query on NA | E [auth HHH] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 58.672 | α = 90 |
| b = 102.374 | β = 90 |
| c = 142.735 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DIALS | data processing |
| Aimless | data scaling |
| Aimless | data reduction |
| PHENIX | phasing |
| Coot | model building |
| REFMAC | refinement |