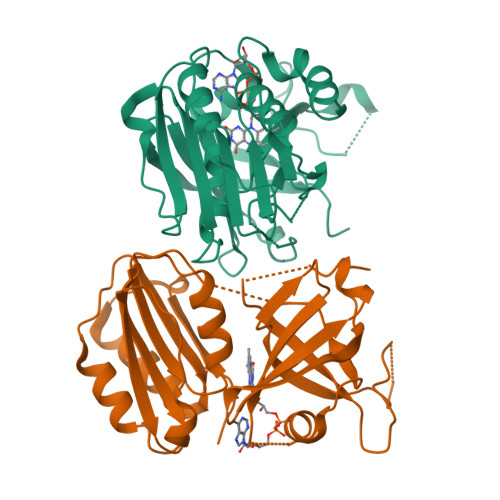
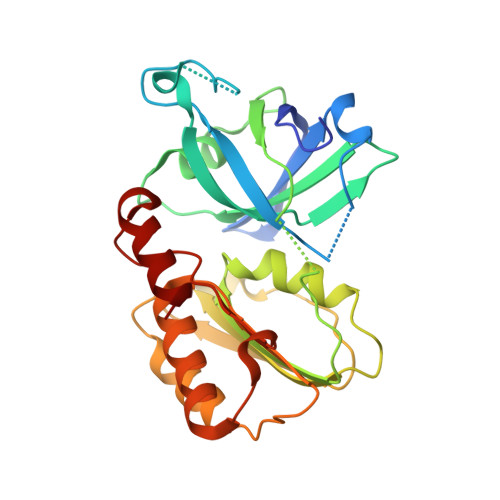
Crystal structure of the Siderophore-interacting protein SIP from Aeromonas hydrophila.
Shang, F., Lan, J., Wang, L., Liu, W., Chen, Y., Chen, J., Ha, N.C., Quan, C., Nam, K.H., Xu, Y.(2019) Biochem Biophys Res Commun 519: 23-28
- PubMed: 31477273
- DOI: https://doi.org/10.1016/j.bbrc.2019.08.085
- Primary Citation of Related Structures:
6K2L - PubMed Abstract:
Siderophores acquire iron from hosts under iron-limiting conditions and play an essential role in the survival of microorganisms. Siderophore-interacting proteins (SIPs) from microbes release iron from the siderophore complex by reducing ferric iron to ferrous iron, but the molecular mechanism of iron reduction remains unclear. To better understand the molecular mechanism of SIPs, we herein report the crystal structure of Aeromonas hydrophila SIP (AhSIP) in complex with flavin adenine dinucleotide (FAD) as a cofactor. AhSIP consists of an N-terminal FAD binding domain and a C-terminal NADH binding domain, which are connected by a linker region. AhSIP showed unique structural differences in the orientation of the cofactor binding lobes when compared with SIP homologs. This study identified a cluster of three basic residues (Lys48, His259 and Arg262) in AhSIP distributed around a potential substrate binding pocket. In addition, AhSIP, containing the NADH binding motif E(L)VL-X 3 -GE, belongs to the group I subfamily. Our results show the diverse cofactor and substrate binding sites of the SIP family.
Organizational Affiliation:
Department of Bioengineering, College of Life Science, Dalian Minzu University, Dalian, 116600, Liaoning, China; Key Laboratory of Biotechnology and Bioresources Utilization (Dalian Minzu University), Ministry of Education, China.