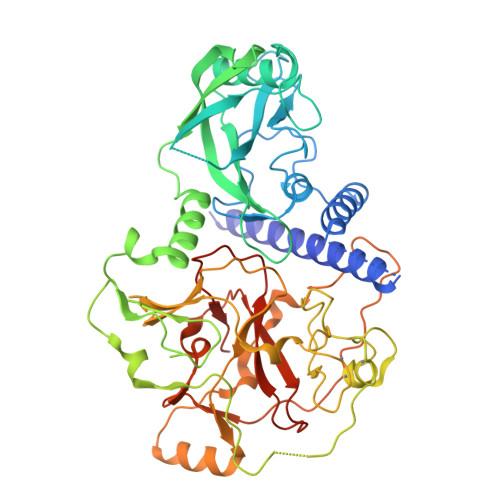
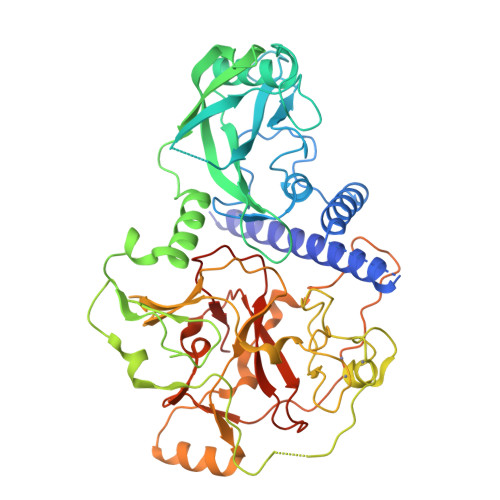
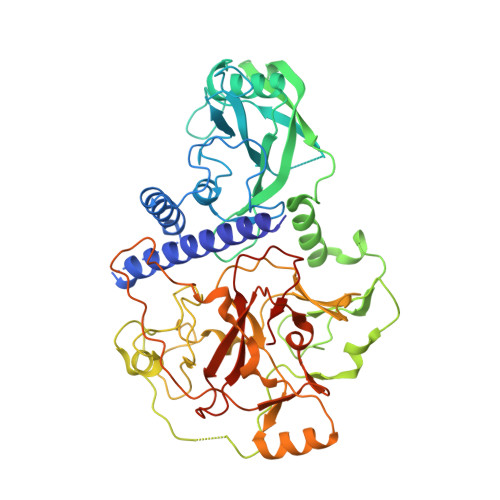
SRA- and SET-domain-containing proteins link RNA polymerase V occupancy to DNA methylation.
Johnson, L.M., Du, J., Hale, C.J., Bischof, S., Feng, S., Chodavarapu, R.K., Zhong, X., Marson, G., Pellegrini, M., Segal, D.J., Patel, D.J., Jacobsen, S.E.(2014) Nature 507: 124-128
- PubMed: 24463519
- DOI: https://doi.org/10.1038/nature12931
- Primary Citation of Related Structures:
4NJ5 - PubMed Abstract:
RNA-directed DNA methylation in Arabidopsis thaliana depends on the upstream synthesis of 24-nucleotide small interfering RNAs (siRNAs) by RNA POLYMERASE IV (Pol IV) and downstream synthesis of non-coding transcripts by Pol V. Pol V transcripts are thought to interact with siRNAs which then recruit DOMAINS REARRANGED METHYLTRANSFERASE 2 (DRM2) to methylate DNA. The SU(VAR)3-9 homologues SUVH2 and SUVH9 act in this downstream step but the mechanism of their action is unknown. Here we show that genome-wide Pol V association with chromatin redundantly requires SUVH2 and SUVH9. Although SUVH2 and SUVH9 resemble histone methyltransferases, a crystal structure reveals that SUVH9 lacks a peptide-substrate binding cleft and lacks a properly formed S-adenosyl methionine (SAM)-binding pocket necessary for normal catalysis, consistent with a lack of methyltransferase activity for these proteins. SUVH2 and SUVH9 both contain SRA (SET- and RING-ASSOCIATED) domains capable of binding methylated DNA, suggesting that they function to recruit Pol V through DNA methylation. Consistent with this model, mutation of DNA METHYLTRANSFERASE 1 (MET1) causes loss of DNA methylation, a nearly complete loss of Pol V at its normal locations, and redistribution of Pol V to sites that become hypermethylated. Furthermore, tethering SUVH9 [corrected] with a zinc finger to an unmethylated site is sufficient to recruit Pol V and establish DNA methylation and gene silencing. These results indicate that Pol V is recruited to DNA methylation through the methyl-DNA binding SUVH2 and SUVH9 proteins, and our mechanistic findings suggest a means for selectively targeting regions of plant genomes for epigenetic silencing.
Organizational Affiliation:
Department of Molecular, Cell and Developmental Biology, University of California at Los Angeles, Los Angeles, CA 90095, USA.