Biochemical properties and atomic resolution structure of a proteolytically processed beta-mannanase from cellulolytic Streptomyces sp. SirexAA-E.
Takasuka, T.E., Acheson, J.F., Bianchetti, C.M., Prom, B.M., Bergeman, L.F., Book, A.J., Currie, C.R., Fox, B.G.(2014) PLoS One 9: e94166-e94166
- PubMed: 24710170
- DOI: https://doi.org/10.1371/journal.pone.0094166
- Primary Citation of Related Structures:
4FK9 - PubMed Abstract:
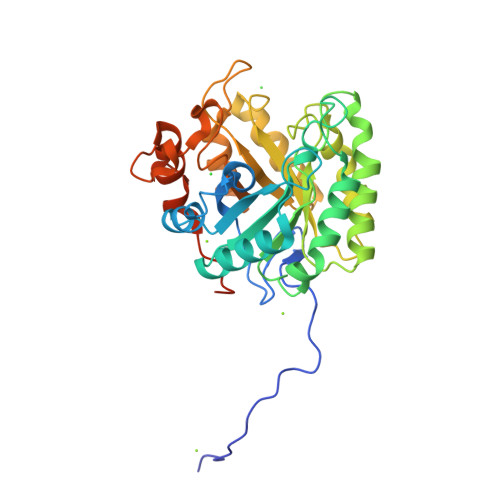
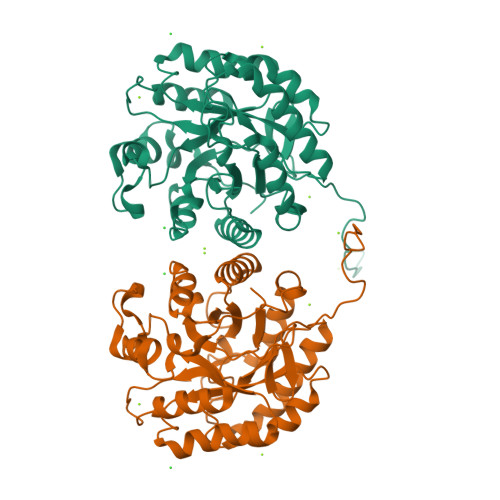
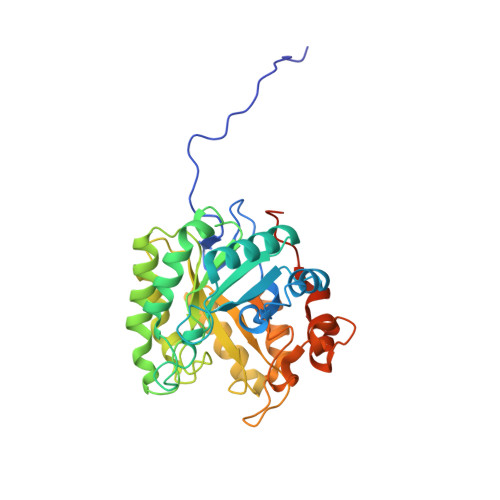
β-Mannanase SACTE_2347 from cellulolytic Streptomyces sp. SirexAA-E is abundantly secreted into the culture medium during growth on cellulosic materials. The enzyme is composed of domains from the glycoside hydrolase family 5 (GH5), fibronectin type-III (Fn3), and carbohydrate binding module family 2 (CBM2). After secretion, the enzyme is proteolyzed into three different, catalytically active variants with masses of 53, 42 and 34 kDa corresponding to the intact protein, loss of the CBM2 domain, or loss of both the Fn3 and CBM2 domains. The three variants had identical N-termini starting with Ala51, and the positions of specific proteolytic reactions in the linker sequences separating the three domains were identified. To conduct biochemical and structural characterizations, the natural proteolytic variants were reproduced by cloning and heterologously expressed in Escherichia coli. Each SACTE_2347 variant hydrolyzed only β-1,4 mannosidic linkages, and also reacted with pure mannans containing partial galactosyl- and/or glucosyl substitutions. Examination of the X-ray crystal structure of the GH5 domain of SACTE_2347 suggests that two loops adjacent to the active site channel, which have differences in position and length relative to other closely related mannanases, play a role in producing the observed substrate selectivity.
Organizational Affiliation:
Great Lakes Bioenergy Research Center, University of Wisconsin -Madison, Madison, Wisconsin, United States of America; Department of Biochemistry, University of Wisconsin -Madison, Madison, Wisconsin, United States of America.