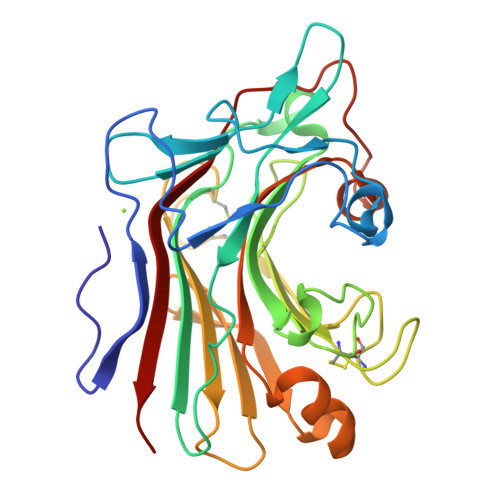
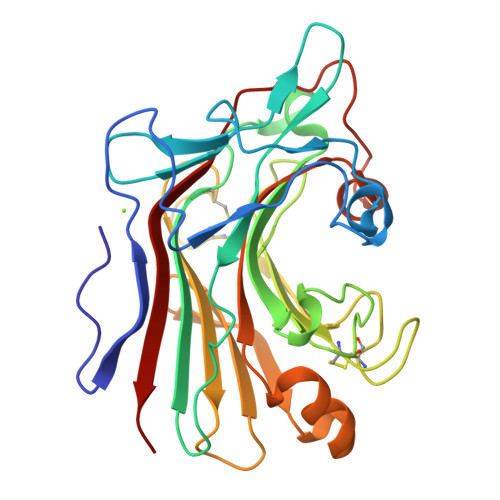
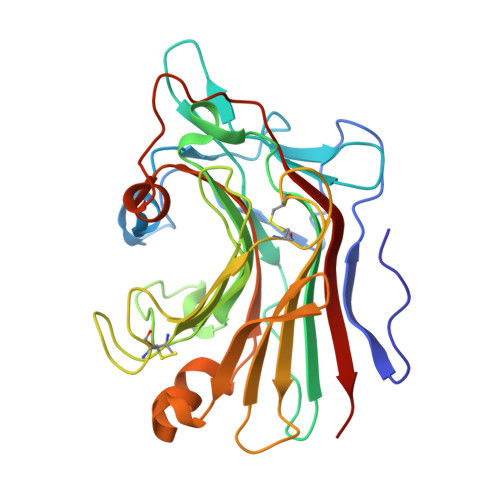
The 1.5 A structure of endo-1,3-beta-glucanase from Streptomyces sioyaensis: evolution of the active-site structure for 1,3-beta-glucan-binding specificity and hydrolysis
Hong, T.-Y., Hsiao, Y.-Y., Meng, M., Li, T.T.(2008) Acta Crystallogr D Biol Crystallogr 64: 964-970
- PubMed: 18703845
- DOI: https://doi.org/10.1107/S0907444908021550
- Primary Citation of Related Structures:
3DGT - PubMed Abstract:
The catalytic domain structure of Streptomyces sioyaensis 1,3-beta-glucanase (278 amino acids), a member of glycosyl hydrolase family 16 (GHF16), was determined to 1.5 A resolution in space group P2(1)2(1)2(1). The enzyme specifically hydrolyzes the glycosidic bond of the 1,3-beta-linked glucan substrate. The overall structure contains two antiparallel six-and seven-stranded beta-sheets stacked in a beta-sandwich jelly-roll motif similar to the fold of GHF16 1,3-1,4-beta-glucanases. The active-site cleft of the enzyme is distinct, with the closure of one end primarily caused by two protruding loop insertions and two key residues, Tyr38 and Tyr134. The current known structures of 1,3-1,4-beta-glucanases and 1,3-beta-glucanase from Nocardiopsis sp., on the other hand, have open-channel active-site clefts that can accommodate six beta-D-glucopyranosyl units. The active-site structure of 1,3-beta-glucanase was compared with those of other homologous structures in order to address the binding and enzymatic specificity for 1,3-beta-linked glucans in Streptomyces. This information could be helpful in the development of specific antifungal agents.
Organizational Affiliation:
Department of Biotechnology, Tajen University, Pingtung 907, Taiwan.