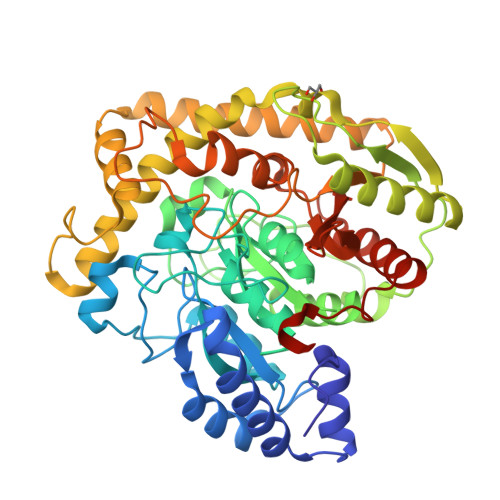
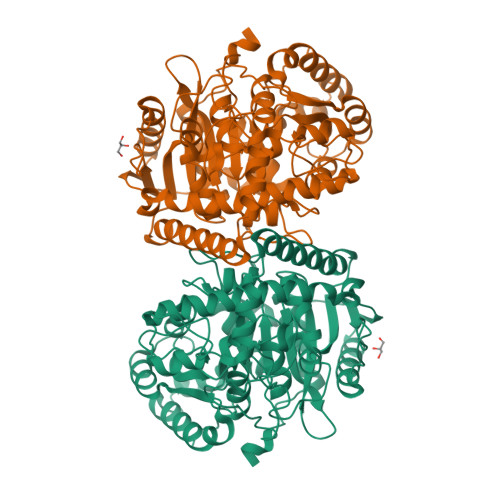
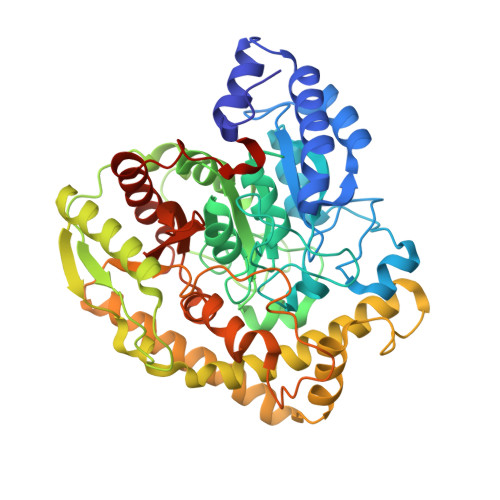
X-ray crystallographic analysis of the 6-aminohexanoate cyclic dimer hydrolase: catalytic mechanism and evolution of an enzyme responsible for nylon-6 byproduct degradation
Yasuhira, K., Shibata, N., Mongami, G., Uedo, Y., Atsumi, Y., Kawashima, Y., Hibino, A., Tanaka, Y., Lee, Y.-H., Kato, D., Takeo, M., Higuchi, Y., Negoro, S.(2010) J Biological Chem 285: 1239-1248
- PubMed: 19889645
- DOI: https://doi.org/10.1074/jbc.M109.041285
- Primary Citation of Related Structures:
3A2P, 3A2Q - PubMed Abstract:
We performed x-ray crystallographic analyses of the 6-aminohexanoate cyclic dimer (Acd) hydrolase (NylA) from Arthrobacter sp., an enzyme responsible for the degradation of the nylon-6 industry byproduct. The fold adopted by the 472-amino acid polypeptide generated a compact mixed alpha/beta fold, typically found in the amidase signature superfamily; this fold was especially similar to the fold of glutamyl-tRNA(Gln) amidotransferase subunit A (z score, 49.4) and malonamidase E2 (z score, 44.8). Irrespective of the high degree of structural similarity to the typical amidase signature superfamily enzymes, the specific activity of NylA for glutamine, malonamide, and indoleacetamide was found to be lower than 0.5% of that for Acd. However, NylA possessed carboxylesterase activity nearly equivalent to the Acd hydrolytic activity. Structural analysis of the inactive complex between the activity-deficient S174A mutant of NylA and Acd, performed at 1.8 A resolution, suggested the following enzyme/substrate interactions: a Ser(174)-cis-Ser(150)-Lys(72) triad constitutes the catalytic center; the backbone N in Ala(171) and Ala(172) are involved in oxyanion stabilization; Cys(316)-S(gamma) forms a hydrogen bond with nitrogen (Acd-N(7)) at the uncleaved amide bond in two equivalent amide bonds of Acd. A single S174A, S150A, or K72A substitution in NylA by site-directed mutagenesis decreased the Acd hydrolytic and esterolytic activities to undetectable levels, indicating that Ser(174)-cis-Ser(150)-Lys(72) is essential for catalysis. In contrast, substitutions at position 316 specifically affected Acd hydrolytic activity, suggesting that Cys(316) is responsible for Acd binding. On the basis of the structure and functional analysis, we discussed the catalytic mechanisms and evolution of NylA in comparison with other Ser-reactive hydrolases.
Organizational Affiliation:
Department of Materials Science and Chemistry, Graduate School of Engineering, University of Hyogo, Hyogo 671-2201, Japan.