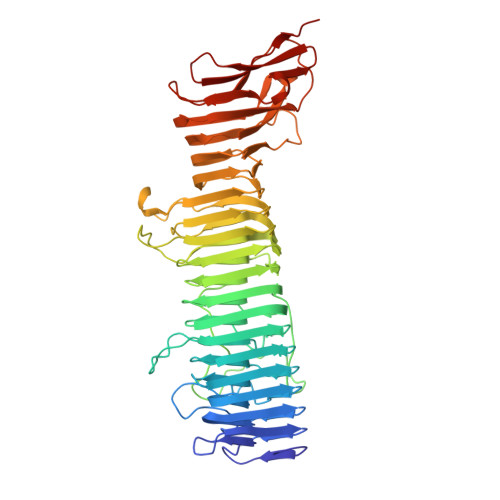
Structure of Bordetella pertussis virulence factor P.69 pertactin.
Emsley, P., Charles, I.G., Fairweather, N.F., Isaacs, N.W.(1996) Nature 381: 90-92
- PubMed: 8609998
- DOI: https://doi.org/10.1038/381090a0
- Primary Citation of Related Structures:
1DAB - PubMed Abstract:
A new generation of whooping-cough vaccines contain P.69 pertactin, a surface-exposed domain of an outer membrane protein expressed by the virulent bacterium Bordetella pertussis. This protein is a virulence factor that mediates adhesion to target mammalian cells, a reaction that is in part mediated by an RGD sequence. The X-ray crystal structure of P.69 pertactin has been determined to 2.5 A. The protein fold consists of a 16-stranded parallel beta-helix with a V-shaped cross-section, and is the largest beta-helix known to date. Several between-strand weakly conserved amino-acid repeats form internal and external ladders. The structure appears as a helix from which several loops protrude, which contain sequence motifs associated with the biological activity of the protein. One particular (GGXXP)5 sequence is located directly after the RGD motif, and may mediate interaction with epithelial cells. The carboxy-terminal region of P.69 pertactin incorporates a (PQP)5 motif loop containing the major immunoprotective epitope.
Organizational Affiliation:
Department of Chemistry, University of Glasgow, UK.