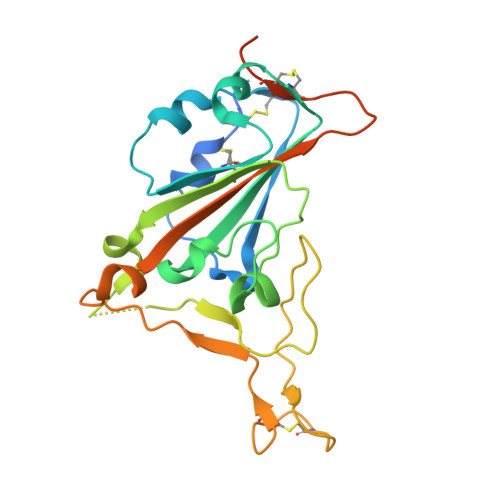
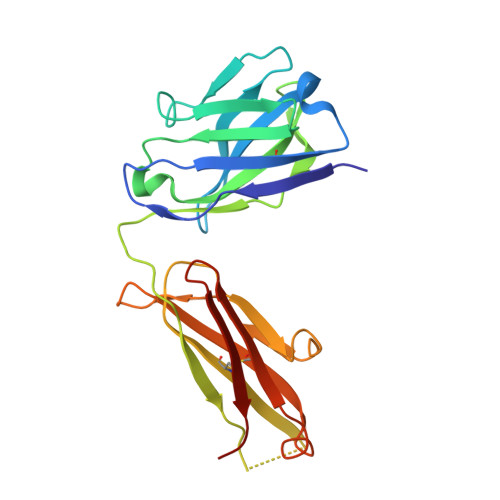
Broadening a SARS-CoV-1-neutralizing antibody for potent SARS-CoV-2 neutralization through directed evolution.
Zhao, F., Yuan, M., Keating, C., Shaabani, N., Limbo, O., Joyce, C., Woehl, J., Barman, S., Burns, A., Tran, Q., Zhu, X., Ricciardi, M., Peng, L., Smith, J., Huang, D., Briney, B., Sok, D., Nemazee, D., Teijaro, J.R., Wilson, I.A., Burton, D.R., Jardine, J.G.(2023) Sci Signal 16: eabk3516-eabk3516
- PubMed: 37582161
- DOI: https://doi.org/10.1126/scisignal.abk3516
- Primary Citation of Related Structures:
8GF2 - PubMed Abstract:
The emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) underscores the need for strategies to rapidly develop neutralizing monoclonal antibodies that can function as prophylactic and therapeutic agents and to help guide vaccine design. Here, we demonstrate that engineering approaches can be used to refocus an existing antibody that neutralizes one virus but not a related virus. Through a rapid affinity maturation strategy, we engineered CR3022, a SARS-CoV-1-neutralizing antibody, to bind to the receptor binding domain of SARS-CoV-2 with >1000-fold increased affinity. The engineered CR3022 neutralized SARS-CoV-2 and provided prophylactic protection from viral challenge in a small animal model of SARS-CoV-2 infection. Deep sequencing throughout the engineering process paired with crystallographic analysis of engineered CR3022 elucidated the molecular mechanisms by which the antibody can accommodate sequence differences in the epitopes between SARS-CoV-1 and SARS-CoV-2. This workflow provides a blueprint for the rapid broadening of neutralization of an antibody from one virus to closely related but resistant viruses.
Organizational Affiliation:
Department of Immunology and Microbiology, Scripps Research Institute, La Jolla, CA 92037, USA.