Design, Synthesis, Biological Activity, and Structural Analysis of Novel Des-C-Ring and Aromatic-D-Ring Analogues of 1 alpha ,25-Dihydroxyvitamin D 3.
Seoane, S., Gogoi, P., Zarate-Ruiz, A., Peluso-Iltis, C., Peters, S., Guiberteau, T., Maestro, M.A., Perez-Fernandez, R., Rochel, N., Mourino, A.(2022) J Med Chem 65: 13112-13124
- PubMed: 36166643
- DOI: https://doi.org/10.1021/acs.jmedchem.2c00900
- Primary Citation of Related Structures:
7ZFG, 7ZFX - PubMed Abstract:
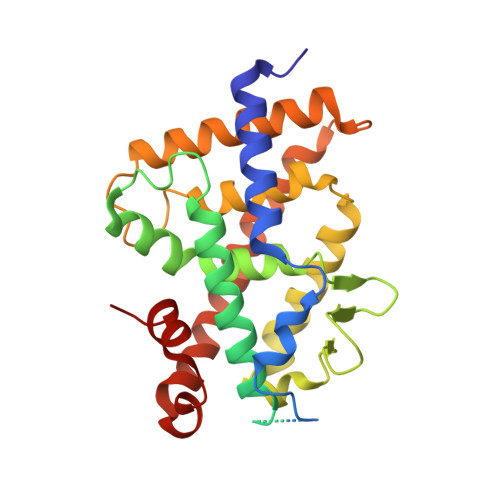
The toxic calcemic effects of the natural hormone 1α,25-dihydroxyvitamin D 3 (1,25D 3 , 1,25-dihydroxycholecalciferol) in the treatment of hyperproliferative diseases demand the development of highly active and noncalcemic vitamin D analogues. We report the development of two highly active and noncalcemic analogues of 1,25D 3 that lack the C-ring and possess an m -phenylene ring that replaces the natural D-ring. The new analogues ( 3a , 3b ) are characterized by an additional six-carbon hydroxylated side chain attached either to the aromatic nucleus or to the triene system. Both compounds were synthesized by the Pd-catalyzed tandem cyclization/cross coupling approach starting from alkyne 6 and diphenol 8 . Key steps include a stereoselective Cu-assisted addition of a Grignard reagent to an aromatic alkyne and a Takai olefination of an aromatic aldehyde. The new compounds are noncalcemic and show transcriptional and antiproliferative activities similar to 1,25D 3 . Structural analysis revealed that they induce a large conformational rearrangement of the vitamin D receptor around helix 6.
Organizational Affiliation:
Department of Physiology-Center for Research in Molecular Medicine and Chronic Diseases (CIMUS), University of Santiago de Compostela, Avda. Barcelona s/n, Santiago de Compostela 15706, Spain.