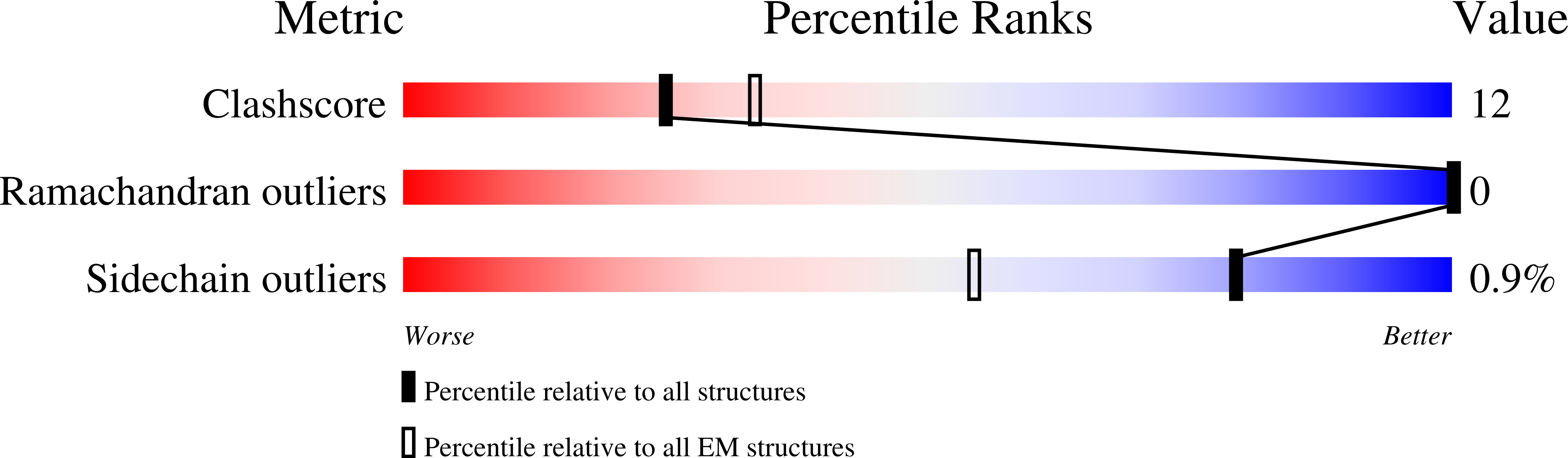A Small-Molecule Oral Agonist of the Human Glucagon-like Peptide-1 Receptor.
Griffith, D.A., Edmonds, D.J., Fortin, J.P., Kalgutkar, A.S., Kuzmiski, J.B., Loria, P.M., Saxena, A.R., Bagley, S.W., Buckeridge, C., Curto, J.M., Derksen, D.R., Dias, J.M., Griffor, M.C., Han, S., Jackson, V.M., Landis, M.S., Lettiere, D., Limberakis, C., Liu, Y., Mathiowetz, A.M., Patel, J.C., Piotrowski, D.W., Price, D.A., Ruggeri, R.B., Tess, D.A.(2022) J Med Chem 65: 8208-8226
- PubMed: 35647711
- DOI: https://doi.org/10.1021/acs.jmedchem.1c01856
- Primary Citation of Related Structures:
7S15 - PubMed Abstract:
Peptide agonists of the glucagon-like peptide-1 receptor (GLP-1R) have revolutionized diabetes therapy, but their use has been limited because they require injection. Herein, we describe the discovery of the orally bioavailable, small-molecule, GLP-1R agonist PF-06882961 (danuglipron). A sensitized high-throughput screen was used to identify 5-fluoropyrimidine-based GLP-1R agonists that were optimized to promote endogenous GLP-1R signaling with nanomolar potency. Incorporation of a carboxylic acid moiety provided considerable GLP-1R potency gains with improved off-target pharmacology and reduced metabolic clearance, ultimately resulting in the identification of danuglipron. Danuglipron increased insulin levels in primates but not rodents, which was explained by receptor mutagensis studies and a cryogenic electron microscope structure that revealed a binding pocket requiring a primate-specific tryptophan 33 residue. Oral administration of danuglipron to healthy humans produced dose-proportional increases in systemic exposure (NCT03309241). This opens an opportunity for oral small-molecule therapies that target the well-validated GLP-1R for metabolic health.
Organizational Affiliation:
Pfizer Worldwide Research, Development, and Medical, Cambridge, Massachusetts 02139, United States.