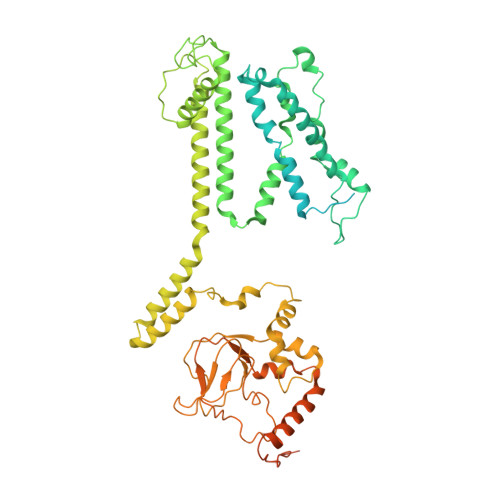
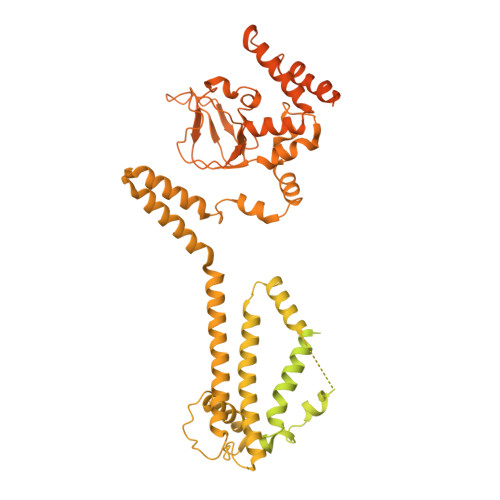
The structure of the native CNGA1/CNGB1 CNG channel from bovine retinal rods.
Barret, D.C.A., Schertler, G.F.X., Benjamin Kaupp, U., Marino, J.(2022) Nat Struct Mol Biol 29: 32-39
- PubMed: 34969975
- DOI: https://doi.org/10.1038/s41594-021-00700-8
- Primary Citation of Related Structures:
7O4H - PubMed Abstract:
In rod photoreceptors of the retina, the cyclic nucleotide-gated (CNG) channel is composed of three CNGA and one CNGB subunits, and it closes in response to light activation to generate an electrical signal that is conveyed to the brain. Here we report the cryo-EM structure of the closed state of the native rod CNG channel isolated from bovine retina. The structure reveals differences between CNGA1 and CNGB1 subunits. Three CNGA1 subunits are tethered at their C terminus by a coiled-coil region. The C-helix in the cyclic nucleotide-binding domain of CNGB1 features a different orientation from that in the three CNGA1 subunits. The arginine residue R994 of CNGB1 reaches into the ionic pathway and blocks the pore, thus introducing an additional gate, which is different from the central hydrophobic gate known from homomeric CNGA channels. These results address the long-standing question of how CNGB1 subunits contribute to the function of CNG channels in visual and olfactory neurons.
Organizational Affiliation:
Laboratory of Biomolecular Research, Paul Scherrer Institut, Villigen, Switzerland.