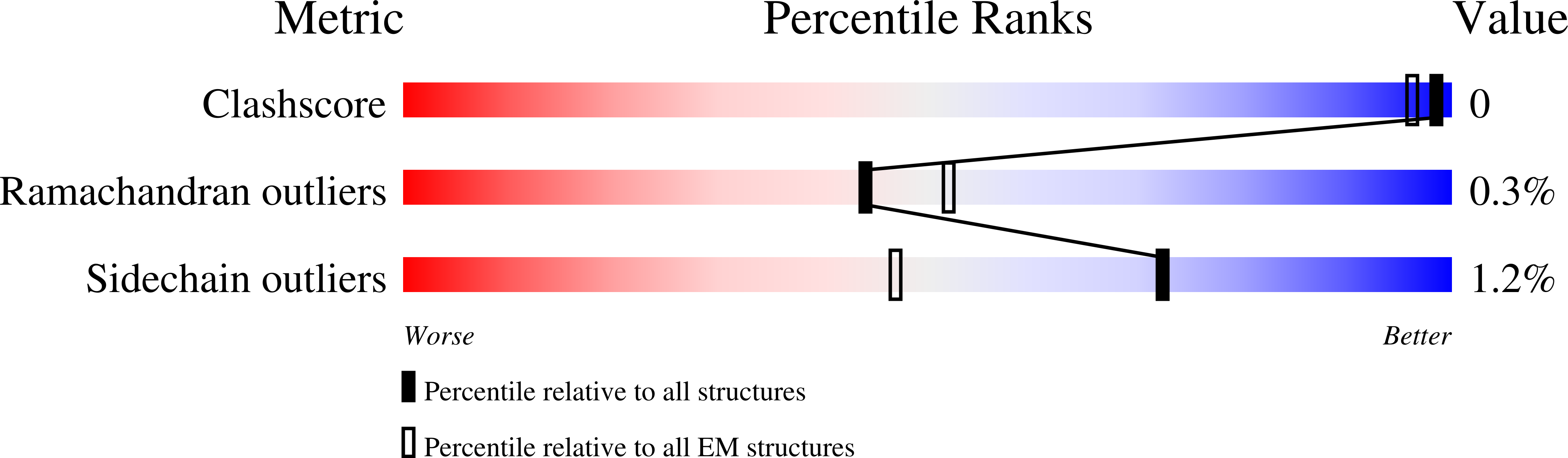Effect of natural mutations of SARS-CoV-2 on spike structure, conformation and antigenicity.
Gobeil, S.M., Janowska, K., McDowell, S., Mansouri, K., Parks, R., Stalls, V., Kopp, M.F., Manne, K., Saunders, K., Edwards, R.J., Haynes, B.F., Henderson, R.C., Acharya, P.(2021) Biorxiv
- PubMed: 33758838
- DOI: https://doi.org/10.1101/2021.03.11.435037
- Primary Citation of Related Structures:
7LWS, 7M0J - PubMed Abstract:
New SARS-CoV-2 variants that have accumulated multiple mutations in the spike (S) glycoprotein enable increased transmission and resistance to neutralizing antibodies. Here, we study the antigenic and structural impacts of the S protein mutations from four variants, one that was involved in transmission between minks and humans, and three that rapidly spread in human populations and originated in the United Kingdom, Brazil or South Africa. All variants either retained or improved binding to the ACE2 receptor. The B.1.1.7 (UK) and B.1.1.28 (Brazil) spike variants showed reduced binding to neutralizing NTD and RBD antibodies, respectively, while the B.1.351 (SA) variant showed reduced binding to both NTD- and RBD-directed antibodies. Cryo-EM structural analyses revealed allosteric effects of the mutations on spike conformations and revealed mechanistic differences that either drive inter-species transmission or promotes viral escape from dominant neutralizing epitopes. Cryo-EM structures reveal changes in SARS-CoV-2 S protein during inter-species transmission or immune evasion.Adaptation to mink resulted in increased ACE2 binding and spike destabilization.B.1.1.7 S mutations reveal an intricate balance of stabilizing and destabilizing effects that impact receptor and antibody binding.E484K mutation in B.1.351 and B.1.1.28 S proteins drives immune evasion by altering RBD conformation.S protein uses different mechanisms to converge upon similar solutions for altering RBD up/down positioning.