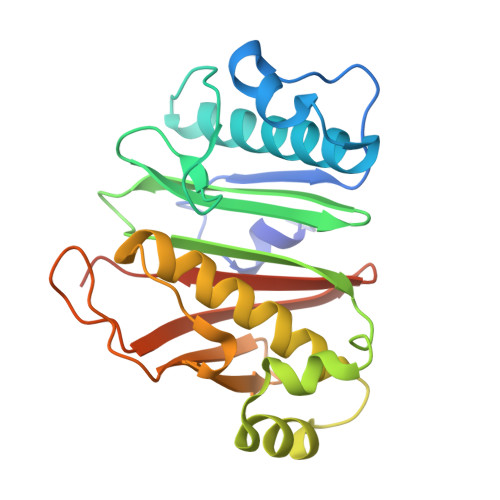
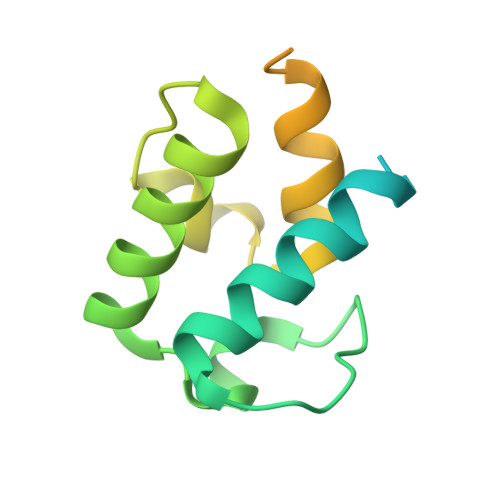
Phosphopantetheinyl transferase binding and inhibition by amidino-urea and hydroxypyrimidinethione compounds.
Carivenc, C., Maveyraud, L., Blanger, C., Ballereau, S., Roy-Camille, C., Nguyen, M.C., Genisson, Y., Guilhot, C., Chalut, C., Pedelacq, J.D., Mourey, L.(2021) Sci Rep 11: 18042-18042
- PubMed: 34508141
- DOI: https://doi.org/10.1038/s41598-021-97197-4
- Primary Citation of Related Structures:
7B4R, 7B4S, 7BCZ, 7BDW - PubMed Abstract:
Owing to their role in activating enzymes essential for bacterial viability and pathogenicity, phosphopantetheinyl transferases represent novel and attractive drug targets. In this work, we examined the inhibitory effect of the aminido-urea 8918 compound against the phosphopantetheinyl transferases PptAb from Mycobacterium abscessus and PcpS from Pseudomonas aeruginosa, two pathogenic bacteria associated with cystic fibrosis and bronchiectasis, respectively. Compound 8918 exhibits inhibitory activity against PptAb but displays no activity against PcpS in vitro, while no antimicrobial activity against Mycobacterium abscessus or Pseudomonas aeruginosa could be detected. X-ray crystallographic analysis of 8918 bound to PptAb-CoA alone and in complex with an acyl carrier protein domain in addition to the crystal structure of PcpS in complex with CoA revealed the structural basis for the inhibition mechanism of PptAb by 8918 and its ineffectiveness against PcpS. Finally, in crystallo screening of potent inhibitors from the National Cancer Institute library identified a hydroxypyrimidinethione derivative that binds PptAb. Both compounds could serve as scaffolds for the future development of phosphopantetheinyl transferases inhibitors.
Organizational Affiliation:
Institut de Pharmacologie et de Biologie Structurale, IPBS, Université de Toulouse, CNRS, UPS, 31077, Toulouse, France.