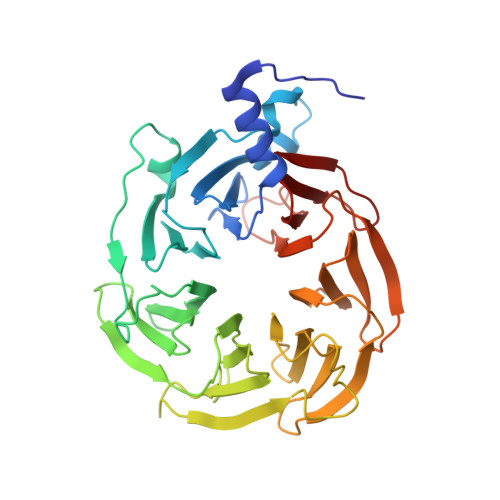
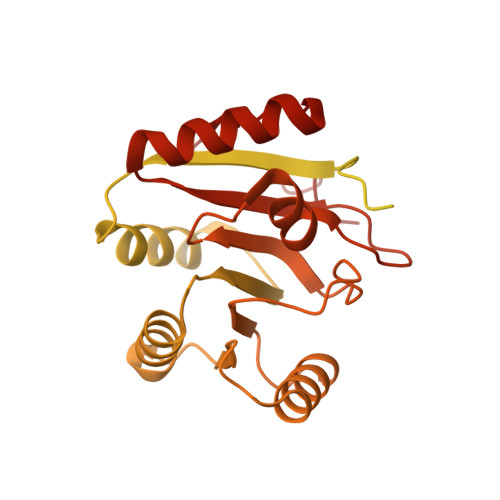
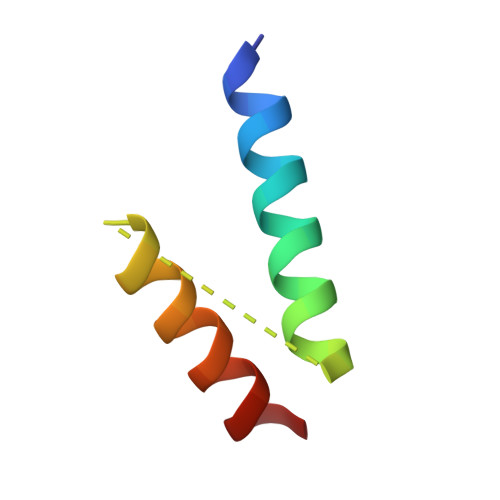
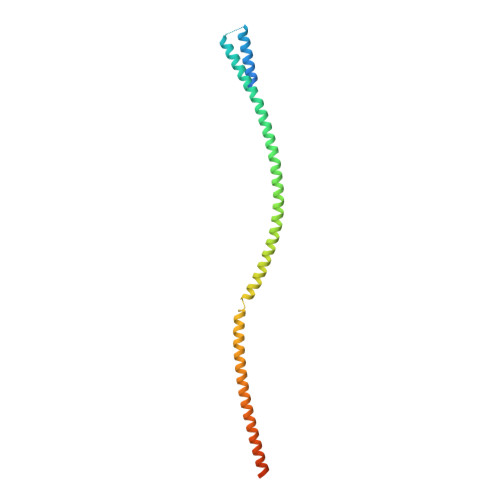Structure of the human core transcription-export complex reveals a hub for multivalent interactions.
Puhringer, T., Hohmann, U., Fin, L., Pacheco-Fiallos, B., Schellhaas, U., Brennecke, J., Plaschka, C.(2020) Elife 9
- PubMed: 33191911
- DOI: https://doi.org/10.7554/eLife.61503
- Primary Citation of Related Structures:
7APK - PubMed Abstract:
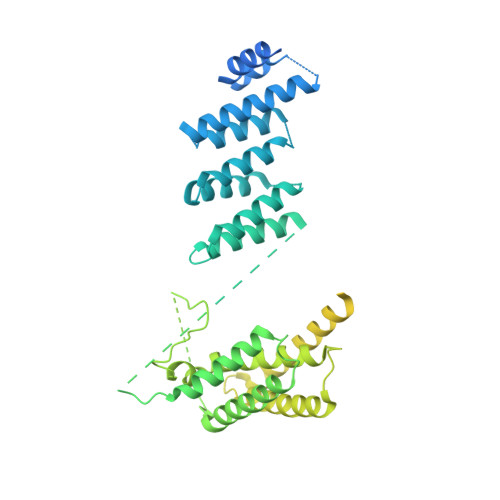
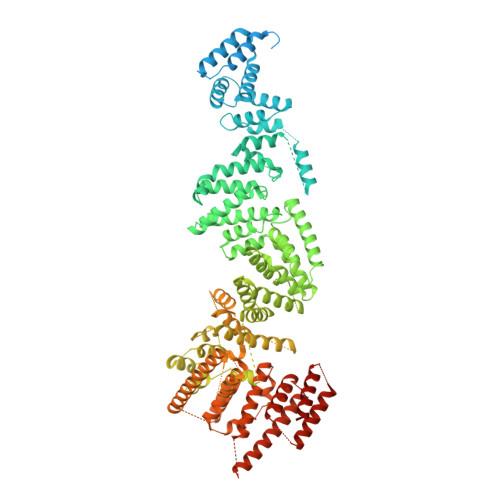
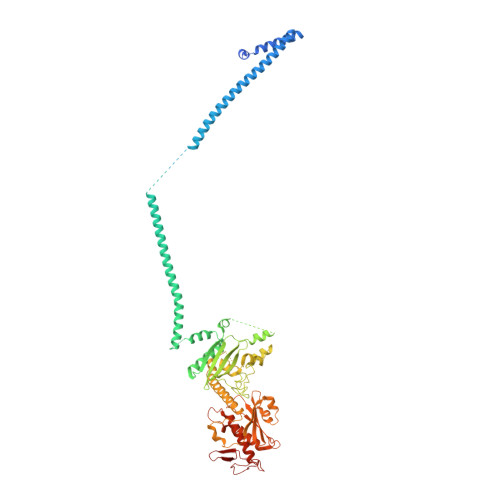
The export of mRNA from nucleus to cytoplasm requires the conserved and essential transcription and export (TREX) complex (THO-UAP56/DDX39B-ALYREF). TREX selectively binds mRNA maturation marks and licenses mRNA for nuclear export by loading the export factor NXF1-NXT1. How TREX integrates these marks and achieves high selectivity for mature mRNA is poorly understood. Here, we report the cryo-electron microscopy structure of the human THO-UAP56/DDX39B complex at 3.3 Å resolution. The seven-subunit THO-UAP56/DDX39B complex multimerizes into a 28-subunit tetrameric assembly, suggesting that selective recognition of mature mRNA is facilitated by the simultaneous sensing of multiple, spatially distant mRNA regions and maturation marks. Two UAP56/DDX39B RNA helicases are juxtaposed at each end of the tetramer, which would allow one bivalent ALYREF protein to bridge adjacent helicases and regulate the TREX-mRNA interaction. Our structural and biochemical results suggest a conserved model for TREX complex function that depends on multivalent interactions between proteins and mRNA.
Organizational Affiliation:
Research Institute of Molecular Pathology (IMP), Vienna BioCenter (VBC), Vienna, Austria.