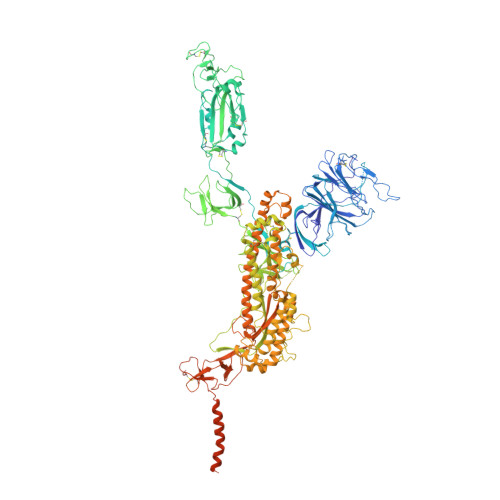
Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM.
Wang, Y., Xu, C., Wang, Y., Hong, Q., Zhang, C., Li, Z., Xu, S., Zuo, Q., Liu, C., Huang, Z., Cong, Y.(2021) Nat Commun 12: 7345-7345
- PubMed: 34930910
- DOI: https://doi.org/10.1038/s41467-021-27350-0
- Primary Citation of Related Structures:
7VX1, 7VX4, 7VX5, 7VX9, 7VXA, 7VXB, 7VXC, 7VXD, 7VXE, 7VXF, 7VXI, 7VXK, 7VXM, 7WEV - PubMed Abstract:
The emergence of SARS-CoV-2 Kappa and Beta variants with enhanced transmissibility and resistance to neutralizing antibodies has created new challenges for the control of the ongoing COVID-19 pandemic. Understanding the structural nature of Kappa and Beta spike (S) proteins and their association with ACE2 is of significant importance. Here we present two cryo-EM structures for each of the Kappa and Beta spikes in the open and open-prone transition states. Compared with wild-type (WT) or G614 spikes, the two variant spikes appear more untwisted/open especially for Beta, and display a considerable population shift towards the open state as well as more pronounced conformational dynamics. Moreover, we capture four conformational states of the S-trimer/ACE2 complex for each of the two variants, revealing an enlarged conformational landscape for the Kappa and Beta S-ACE2 complexes and pronounced population shift towards the three RBDs up conformation. These results implicate that the mutations in Kappa and Beta may modify the kinetics of receptor binding and viral fusion to improve virus fitness. Combined with biochemical analysis, our structural study shows that the two variants are enabled to efficiently interact with ACE2 receptor despite their sensitive ACE2 binding surface is modified to escape recognition by some potent neutralizing MAbs. Our findings shed new light on the pathogenicity and immune evasion mechanism of the Beta and Kappa variants.
Organizational Affiliation:
State Key Laboratory of Molecular Biology, National Center for Protein Science Shanghai, Shanghai Institute of Biochemistry and Cell Biology, Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences, 200031, Shanghai, China.