Mechanisms of the Antiproliferative and Antitumor Activity of Novel Telomerase-Carbonic Anhydrase Dual-Hybrid Inhibitors.
Plyasova, A.A., Berrino, E., Khan, I.I., Veselovsky, A.V., Pokrovsky, V.S., Angeli, A., Ferraroni, M., Supuran, C.T., Pokrovskaya, M.V., Alexandrova, S.S., Gladilina, Y.A., Sokolov, N.N., Hilal, A., Carta, F., Zhdanov, D.D.(2021) J Med Chem 64: 11432-11444
- PubMed: 34283610
- DOI: https://doi.org/10.1021/acs.jmedchem.1c00756
- Primary Citation of Related Structures:
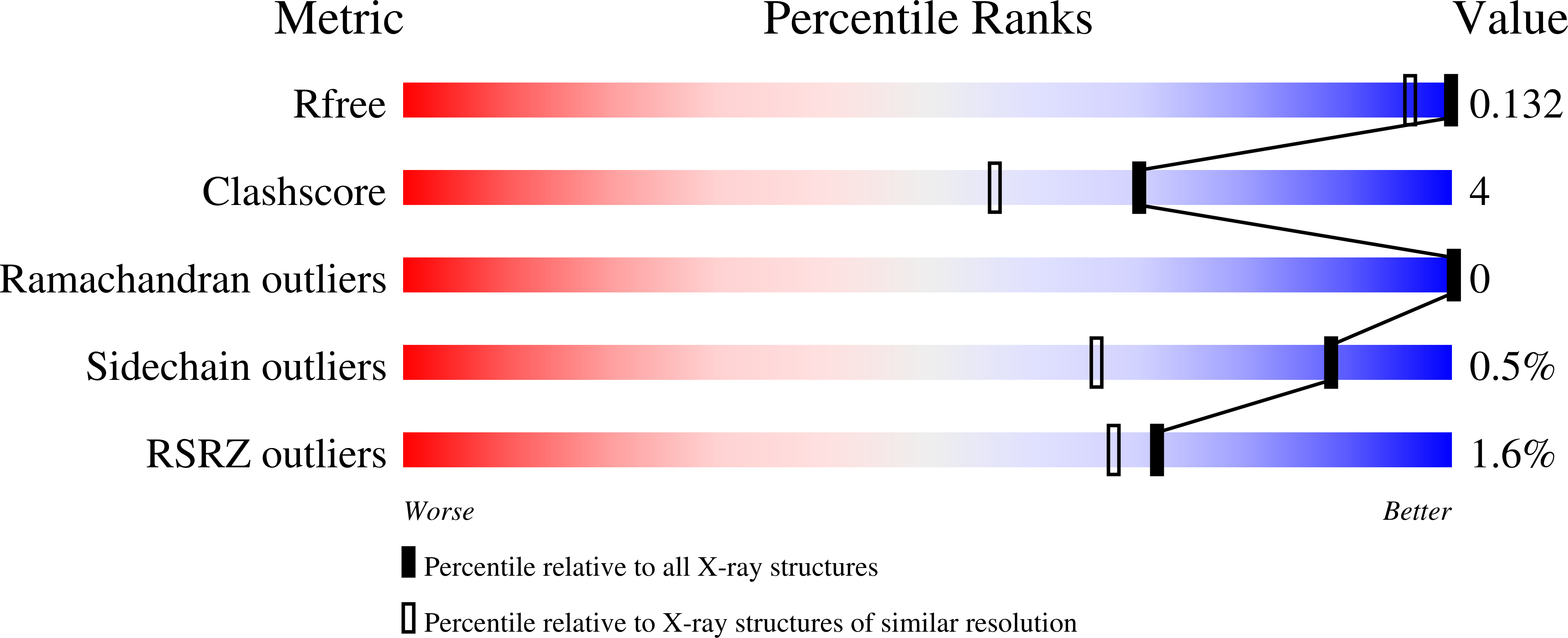
6YPW, 7NH6 - PubMed Abstract:
Human (h) telomerase (TL; EC 2.7.7.49) plays a key role in sustaining cancer cells by means of elongating telomeric repeats at the 3' ends of chromosomes. Since TL-inhibitor (TI) stand-alone cancer therapy has been proven to be remarkably challenging, a polypharmacological approach represents a valid alternative. Here we consider a series of compounds able to inhibit both hTL and the tumor-associated carbonic anhydrases (CAs; EC 4.2.1.1) IX and XII. Compounds 7 and 9 suppressed hTL activity in both cell lysates and human colon cancer cell lines, and prolonged incubation with either 7 or 9 resulted in telomere shortening, cell cycle arrest, replicative senescence, and apoptosis. Enzyme kinetics showed that 7 and 9 are mixed-type inhibitors of the binding of DNA primers and deoxynucleoside triphosphate (dNTP) to the TL catalytic subunit hTERT, which is in agreement with docking experiments. Compound 9 showed antitumor activity in Colo-205 mouse xenografts and suppressed telomerase activity by telomere reduction.
Organizational Affiliation:
Institute of Biomedical Chemistry, Pogodinskaya Street 10/8, Moscow 119121, Russia.