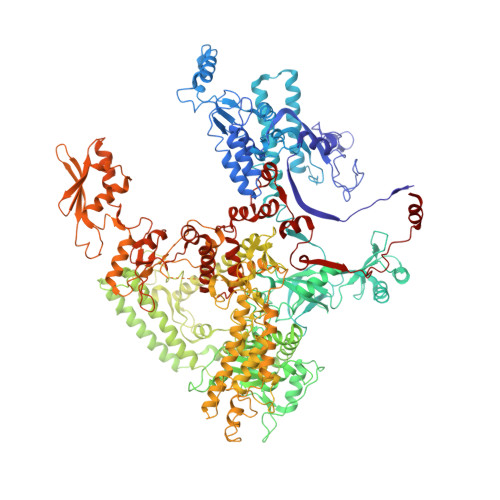
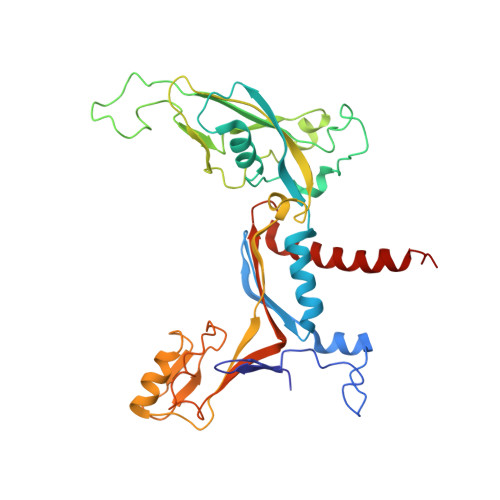
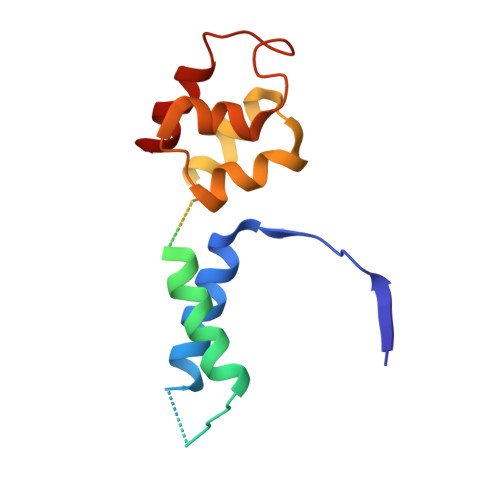
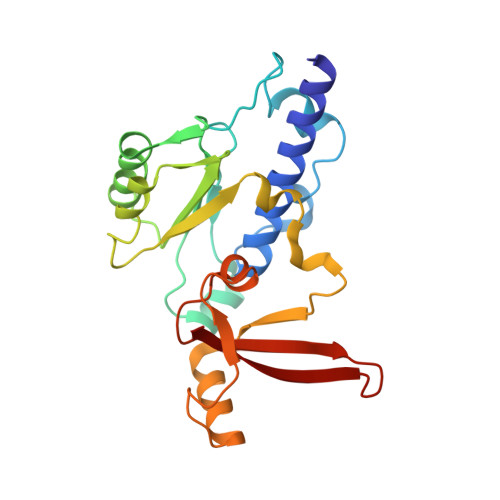
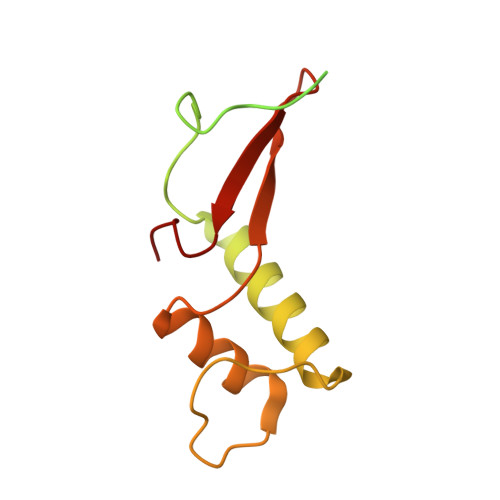
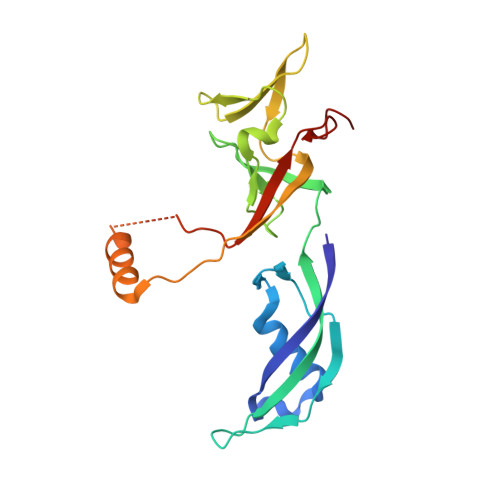
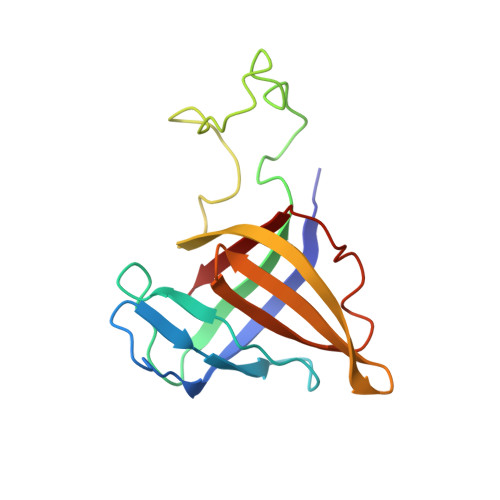
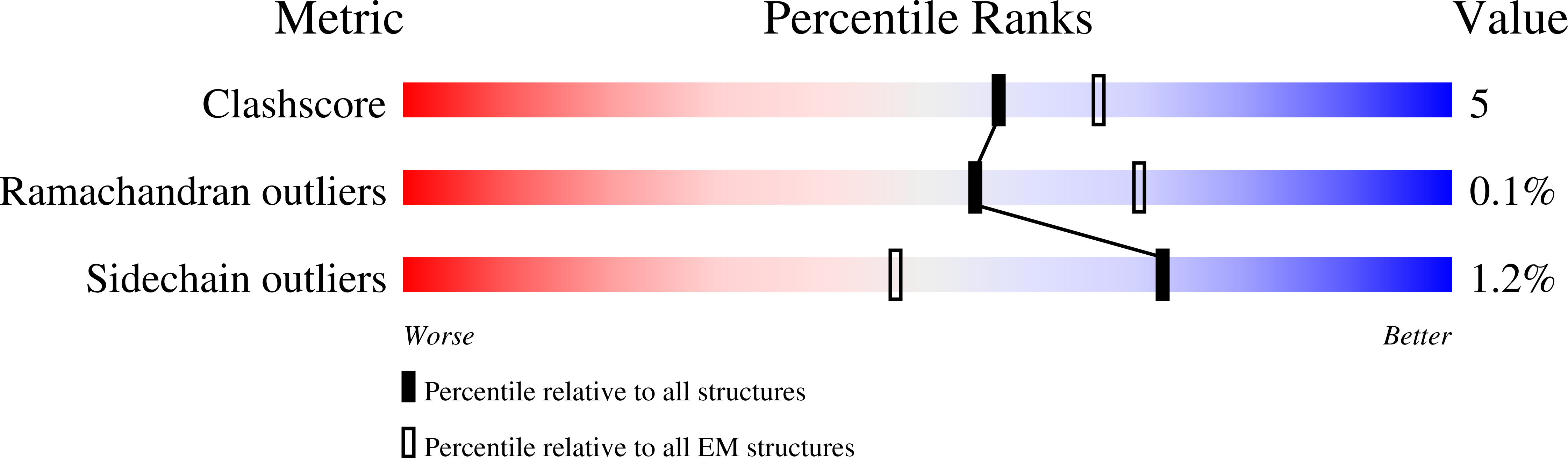Structural basis for RNA polymerase III transcription repression by Maf1.
Vorlander, M.K., Baudin, F., Moir, R.D., Wetzel, R., Hagen, W.J.H., Willis, I.M., Muller, C.W.(2020) Nat Struct Mol Biol 27: 229-232
- PubMed: 32066962
- DOI: https://doi.org/10.1038/s41594-020-0383-y
- Primary Citation of Related Structures:
6TUT - PubMed Abstract:
Maf1 is a conserved inhibitor of RNA polymerase III (Pol III) that influences phenotypes ranging from metabolic efficiency to lifespan. Here, we present a 3.3-Å-resolution cryo-EM structure of yeast Maf1 bound to Pol III, establishing that Maf1 sequesters Pol III elements involved in transcription initiation and binds the mobile C34 winged helix 2 domain, sealing off the active site. The Maf1 binding site overlaps with that of TFIIIB in the preinitiation complex.
Organizational Affiliation:
Structural and Computational Biology Unit, European Molecular Biology Laboratory (EMBL), Heidelberg, Germany.