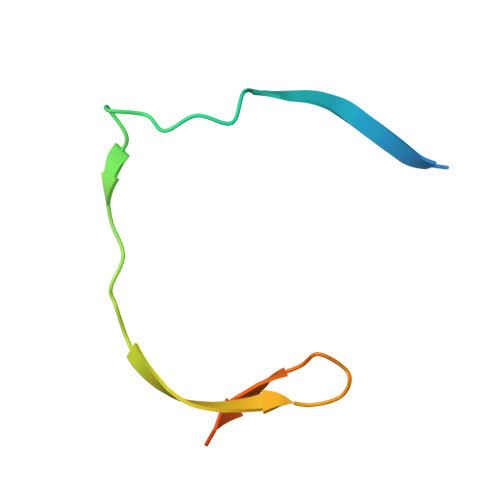
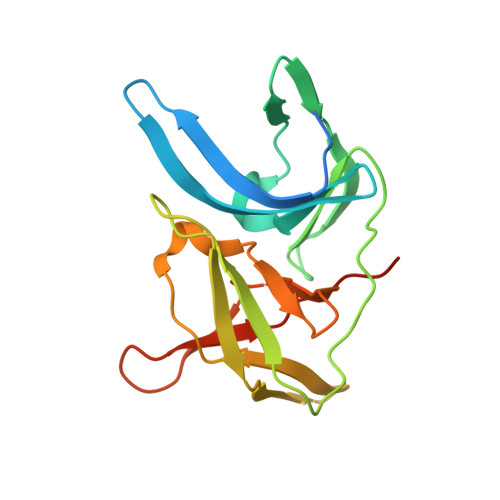
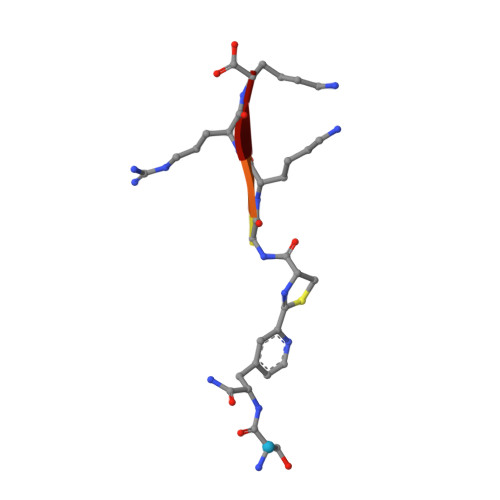
Biocompatible Macrocyclization between Cysteine and 2-Cyanopyridine Generates Stable Peptide Inhibitors.
Nitsche, C., Onagi, H., Quek, J.P., Otting, G., Luo, D., Huber, T.(2019) Org Lett 21: 4709-4712
- PubMed: 31188009
- DOI: https://doi.org/10.1021/acs.orglett.9b01545
- Primary Citation of Related Structures:
6JPW - PubMed Abstract:
Peptides featuring an N-terminal cysteine residue and the unnatural amino acid 3-(2-cyano-4-pyridyl)alanine (Cpa) cyclize spontaneously in aqueous solution at neutral pH. Cpa is readily available and easily introduced into peptides using standard solid-phase peptide synthesis. The reaction is orthogonal to all proteinogenic amino acids, including cysteine residues that are not at the N-terminus. A substrate peptide of the Zika virus NS2B-NS3 protease cyclized in this way produced an inhibitor of high affinity and proteolytic stability.
Organizational Affiliation:
Research School of Chemistry , Australian National University , Canberra , ACT 2601 , Australia.