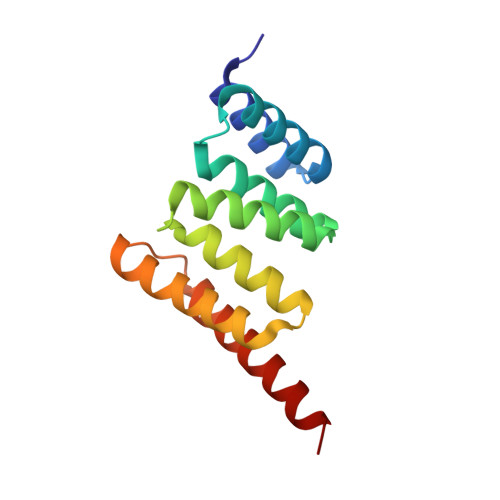
Specificity for latent C termini links the E3 ubiquitin ligase CHIP to caspases.
Ravalin, M., Theofilas, P., Basu, K., Opoku-Nsiah, K.A., Assimon, V.A., Medina-Cleghorn, D., Chen, Y.F., Bohn, M.F., Arkin, M., Grinberg, L.T., Craik, C.S., Gestwicki, J.E.(2019) Nat Chem Biol 15: 786-794
- PubMed: 31320752
- DOI: https://doi.org/10.1038/s41589-019-0322-6
- Primary Citation of Related Structures:
6EFK, 6NSV - PubMed Abstract:
Protein-protein interactions between E3 ubiquitin ligases and protein termini help shape the proteome. These interactions are sensitive to proteolysis, which alters the ensemble of cellular N and C termini. Here we describe a mechanism wherein caspase activity reveals latent C termini that are then recognized by the E3 ubiquitin ligase CHIP. Using expanded knowledge of CHIP's binding specificity, we predicted hundreds of putative interactions arising from caspase activity. Subsequent validation experiments confirmed that CHIP binds the latent C termini at tau D421 and caspase-6 D179 . CHIP binding to tau D421 , but not tau FL , promoted its ubiquitination, while binding to caspase-6 D179 mediated ubiquitin-independent inhibition. Given that caspase activity generates tau D421 in Alzheimer's disease (AD), these results suggested a concise model for CHIP regulation of tau homeostasis. Indeed, we find that loss of CHIP expression in AD coincides with the accumulation of tau D421 and caspase-6 D179 . These results illustrate an unanticipated link between caspases and protein homeostasis.
Organizational Affiliation:
Department of Pharmaceutical Chemistry, University of California at San Francisco, San Francisco, CA, USA.