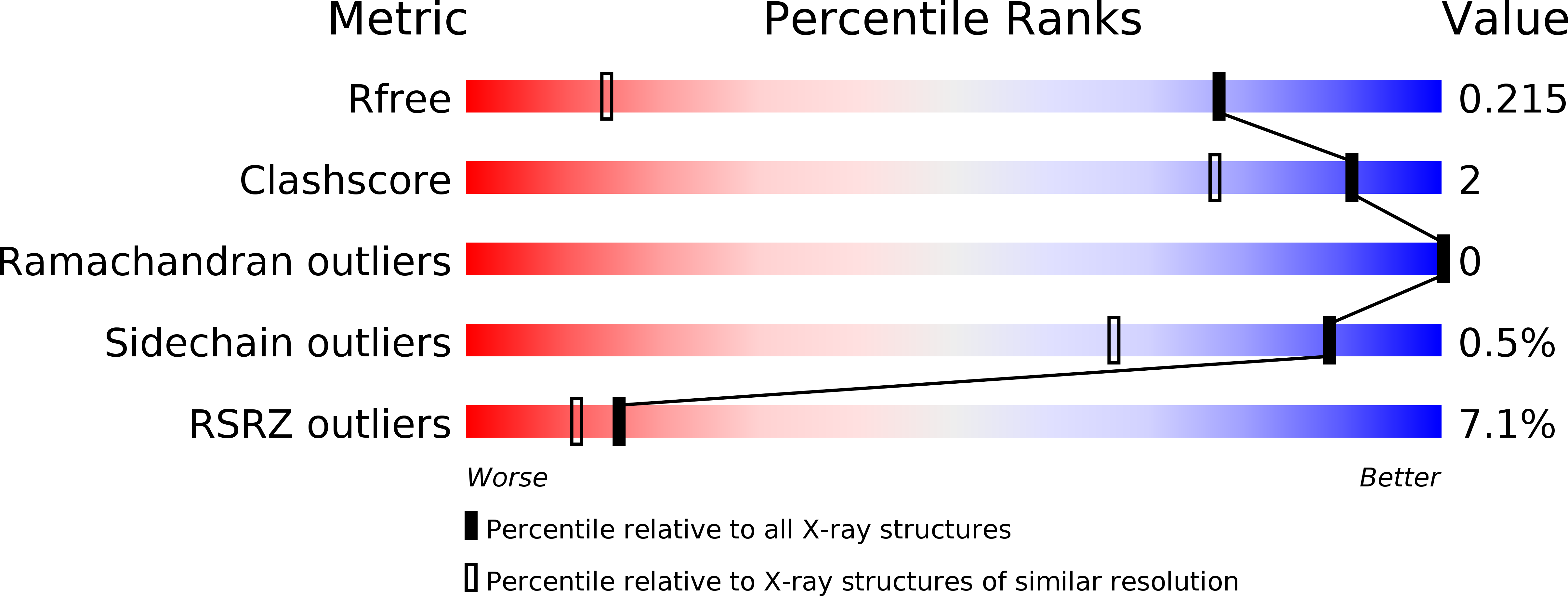
Discovery of potent, highly selective covalent irreversible BTK inhibitors from a fragment hit.
Qiu, H., Liu-Bujalski, L., Caldwell, R.D., Follis, A.V., Gardberg, A., Goutopoulos, A., Grenningloh, R., Head, J., Johnson, T., Jones, R., Mochalkin, I., Morandi, F., Neagu, C., Sherer, B.(2018) Bioorg Med Chem Lett 28: 2939-2944
- PubMed: 30122225
- DOI: https://doi.org/10.1016/j.bmcl.2018.07.008
- Primary Citation of Related Structures:
6DI3, 6DI5, 6DI9 - PubMed Abstract:
Bruton's Tyrosine Kinase (BTK) is a member of the TEC kinase family that is expressed in cells of hematopoietic lineage (e.g., in B cells, macrophages, monocytes, and mast cells). Small molecule covalent irreversible BTK inhibitor targeting Cys481 within the ATP-binding pocket, for example ibrutinib, has been applied in the treatment of B-cell malignancies. Starting from a fragment hit, we discovered a novel series of potent covalent irreversible BTK inhibitors that occupy selectivity pocket of the active site of the BTK kinase domain. Guided by X-ray structures and a fragment-based drug design (FBDD) approach, we generated molecules showing comparable cellular potency to ibrutinib and higher kinome selectivity against undesirable off-targets like EGFR.
Organizational Affiliation:
EMD Serono Research & Development Institute, Inc., 45A Middlesex Turnpike, Billerica 01821, MA, USA. Electronic address: hui.qiu@emdserono.com.