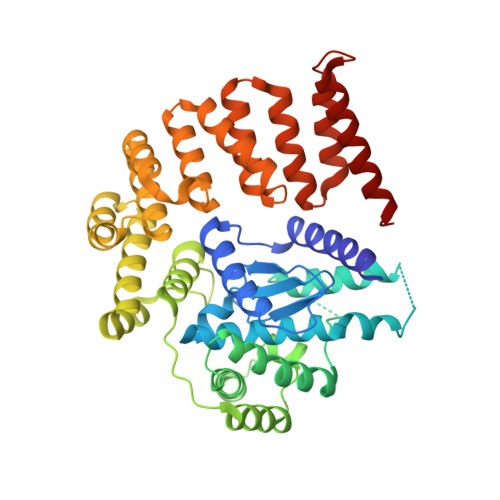
Structural Basis for the Function of the beta-Barrel Assembly-Enhancing Protease BepA.
Shahrizal, M., Daimon, Y., Tanaka, Y., Hayashi, Y., Nakayama, S., Iwaki, S., Narita, S.I., Kamikubo, H., Akiyama, Y., Tsukazaki, T.(2019) J Mol Biol 431: 625-635
- PubMed: 30521812
- DOI: https://doi.org/10.1016/j.jmb.2018.11.024
- Primary Citation of Related Structures:
6AIT - PubMed Abstract:
The β-barrel assembly machinery (BAM) complex mediates the assembly of β-barrel membrane proteins in the outer membrane. BepA, formerly known as YfgC, interacts with the BAM complex and functions as a protease/chaperone for the enhancement of the assembly and/or degradation of β-barrel membrane proteins. To elucidate the molecular mechanism underlying the dual functions of BepA, its full-length three-dimensional structure is needed. Here, we report the crystal structure of full-length BepA at 2.6-Å resolution. BepA possesses an N-terminal protease domain and a C-terminal tetratricopeptide repeat domain, which interact with each other. Domain cross-linking by structure-guided introduction of disulfide bonds did not affect the activities of BepA in vivo, suggesting that the function of this protein does not involve domain rearrangement. The full-length BepA structure is compatible with the previously proposed docking model of BAM complex and tetratricopeptide repeat domain of BepA.
Organizational Affiliation:
Nara Institute of Science and Technology, Ikoma, Nara 630-0192, Japan.