Crystal structure of the reductase (C1) component of p-hydroxyphenylacetate 3-hydroxylase (HPAH) from Acinetobacter baumannii
Oonanant, W., Phongsak, T., Sucharitakul, J., Chaiyen, P., Yuvaniyama, J.To be published.
Experimental Data Snapshot
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| p-hydroxyphenylacetate 3-hydroxylase, reductase component | 315 | Acinetobacter baumannii | Mutation(s): 0 Gene Names: C1-hpah EC: 1.5.1.36 | 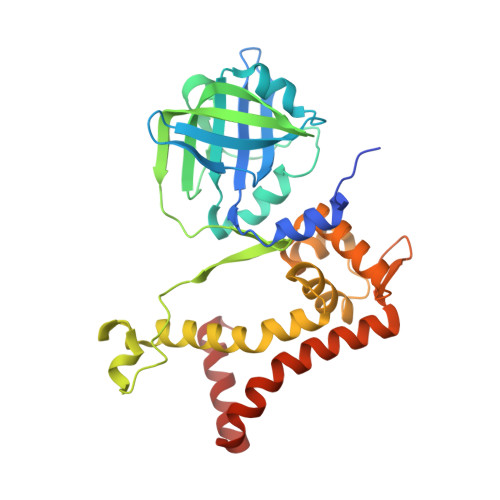 | |
UniProt | |||||
Find proteins for Q6Q271 (Acinetobacter baumannii) Explore Q6Q271 Go to UniProtKB: Q6Q271 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q6Q271 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FMN Query on FMN | C [auth A], E [auth B] | FLAVIN MONONUCLEOTIDE C17 H21 N4 O9 P FVTCRASFADXXNN-SCRDCRAPSA-N |  | ||
| ACT Query on ACT | D [auth A], F [auth B] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 47.992 | α = 90 |
| b = 59.9 | β = 90 |
| c = 212.3 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| CNS | phasing |
| Coot | model building |
| PHENIX | refinement |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Synchrotron Light Research Institute | Thailand | 2-2549/LS02 |
| Thailand | -- |