Production of an active form of vitamin D2 by genetically engineered CYP105A1
Yasuda, K., Yogo, Y., Sugimoto, H., Mano, H., Takita, T., Ohta, M., Kamakura, M., Ikushiro, S., Yasukawa, K., Shiro, Y., Sakaki, T.(2017) Biochem Biophys Res Commun 486: 336-341
- PubMed: 28302483
- DOI: https://doi.org/10.1016/j.bbrc.2017.03.040
- Primary Citation of Related Structures:
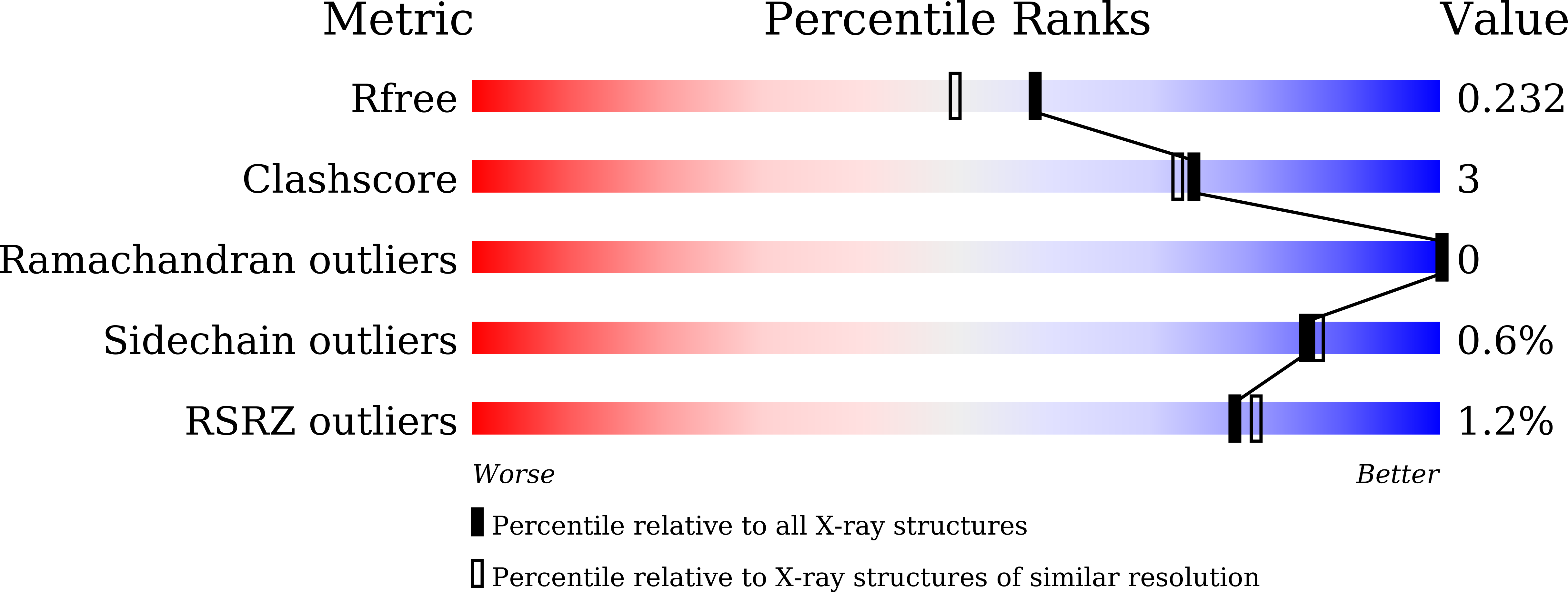
5X7E - PubMed Abstract:
Our previous studies revealed that CYP105A1 can convert vitamin D 3 (VD3) to its active form, 1α,25-dihydroxyvitamin D 3 (1,25D3). Site-directed mutagenesis of CYP105A1 based on its crystal structure dramatically enhanced its activity; the activity of double variants R73A/R84A and R73A/R84V was more than 100-fold higher than that of the wild type of CYP105A1. In contrast, these variants had a low ability to convert vitamin D 2 (VD2) to 1α,25-dihydroxyvitamin D 2 (1,25D2), whereas they catalyzed the sequential hydroxylation at positions C25 and C26 to produce 25,26D2. A comparison of the docking models of 25D2 and 25D3 into the substrate-binding pocket of R73A/R84A suggests that the side chain of the Met239 inhibits the binding of 25D2 for 1α-hydroxylation. Therefore, the Met239 residue of R73A/R84A was substituted for Ala. As expected, the triple variant R73A/R84A/M239A showed a 22-fold higher 1α-hydroxylation activity towards 25D2. To the best of our knowledge, this is the first report on the generation of microbial cytochrome P450 that converts VD2 to 1,25D2 via 25D2.
Organizational Affiliation:
Department of Biotechnology, Faculty of Engineering, Toyama Prefectural University, 5180 Kurokawa, Imizu, Toyama 939-0398, Japan.