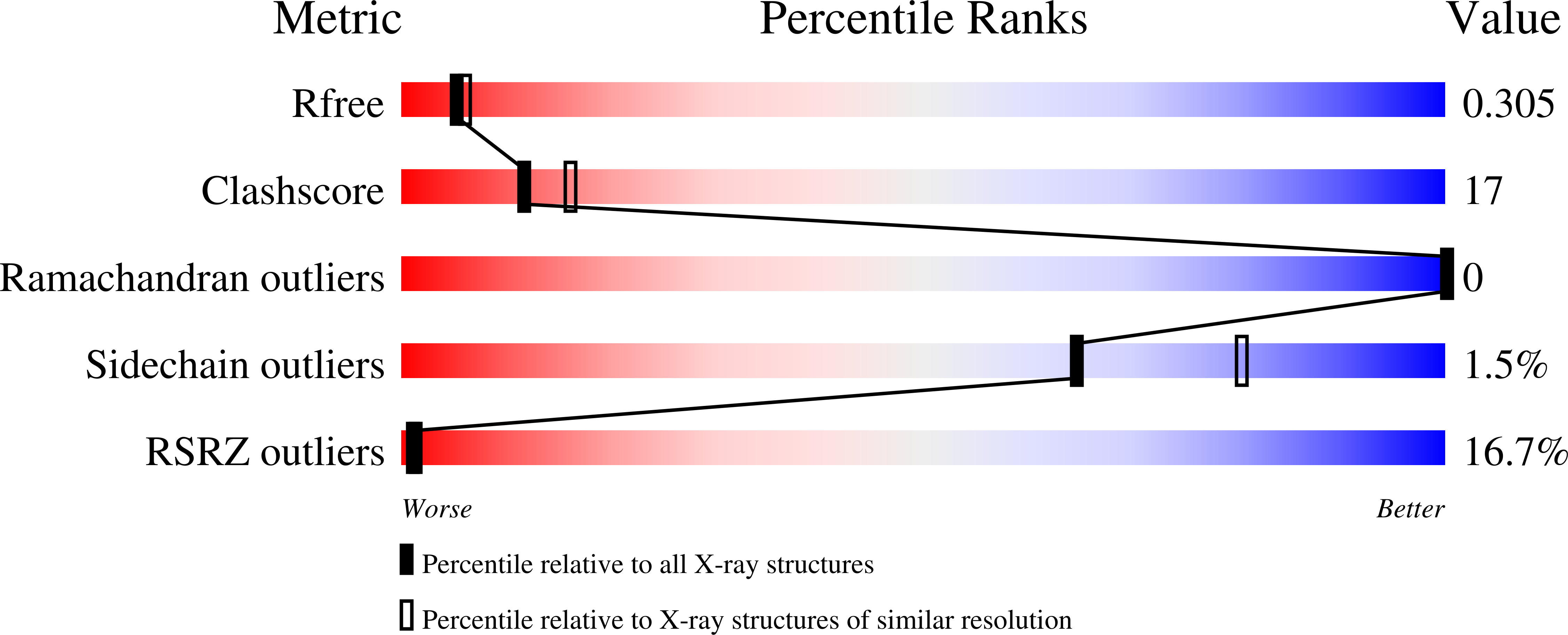
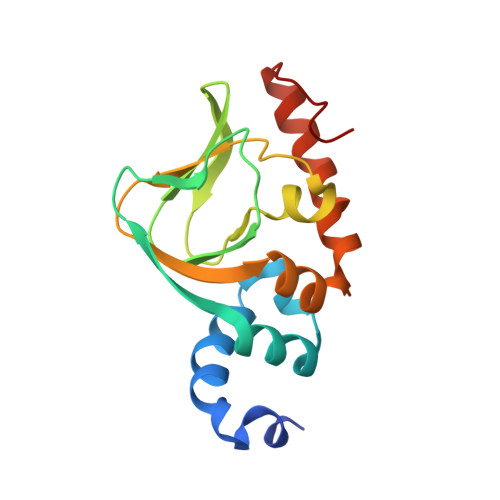
Disrupting the Allosteric Interaction between the Plasmodium falciparum cAMP-dependent Kinase and Its Regulatory Subunit.
Littler, D.R., Bullen, H.E., Harvey, K.L., Beddoe, T., Crabb, B.S., Rossjohn, J., Gilson, P.R.(2016) J Biol Chem 291: 25375-25386
- PubMed: 27738107
- DOI: https://doi.org/10.1074/jbc.M116.750174
- Primary Citation of Related Structures:
5K8S, 5KBF, 5T3N - PubMed Abstract:
The ubiquitous second messenger cAMP mediates signal transduction processes in the malarial parasite that regulate host erythrocyte invasion and the proliferation of merozoites. In Plasmodium falciparum, the central receptor for cAMP is the single regulatory subunit (R) of protein kinase A (PKA). To aid the development of compounds that can selectively dysregulate parasite PKA signaling, we solved the structure of the PKA regulatory subunit in complex with cAMP and a related analogue that displays antimalarial activity, (S p )-2-Cl-cAMPS. Prior to signaling, PKA-R holds the kinase's catalytic subunit (C) in an inactive state by exerting an allosteric inhibitory effect. When two cAMP molecules bind to PKA-R, they stabilize a structural conformation that facilitates its dissociation, freeing PKA-C to phosphorylate downstream substrates such as apical membrane antigen 1. Although PKA activity was known to be necessary for erythrocytic proliferation, we show that uncontrolled induction of PKA activity using membrane-permeable agonists is equally disruptive to growth.
Organizational Affiliation:
From the Infection and Immunity Program and Department of Biochemistry and Molecular Biology, Biomedicine Discovery Institute, and.