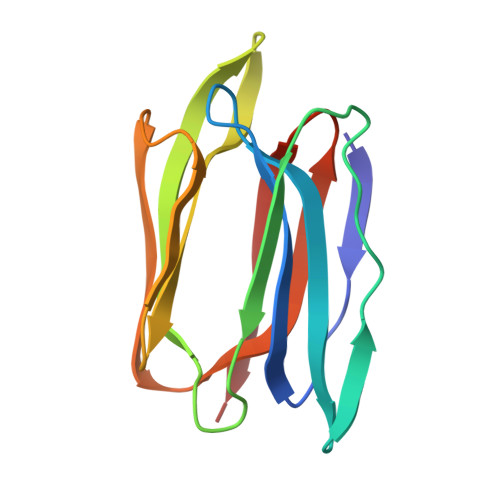
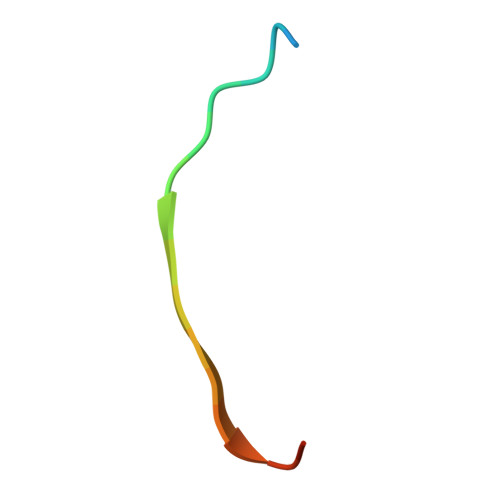
Effect of linkage on the location of reducing and nonreducing sugars bound to jacalin.
Abhinav, K.V., Sharma, K., Surolia, A., Vijayan, M.(2016) IUBMB Life 68: 971-979
- PubMed: 27808459
- DOI: https://doi.org/10.1002/iub.1572
- Primary Citation of Related Structures:
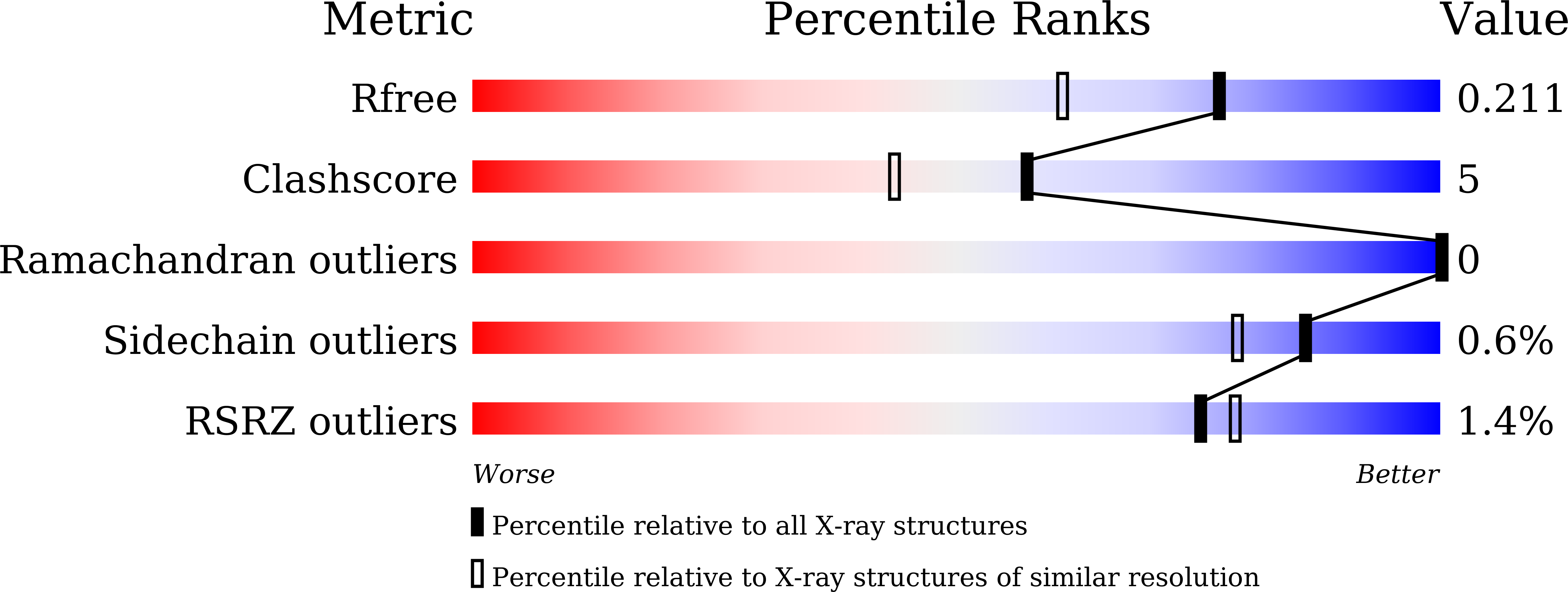
5J51, 5JM1 - PubMed Abstract:
The crystal structures of jacalin complexed with Gal α-(1,4) Gal and Gal α-(1,3) Gal β-(1,4) Gal have been determined with the primary objective of exploring the effect of linkage on the location of reducing and non-reducing sugars in the extended binding site of the lectin, an issue which has not been studied thoroughly. Contrary to the earlier surmise based on simple steric considerations, the two structures demonstrate that α-linked sugars can bind to jacalin with nonreducing sugar at the primary binding site. This is made possible substantially on account of the hitherto underestimated plasticity of a non-polar region of the extended binding site. Modeling studies involving conformational search and energy minimization, along with available crystallographic and thermodynamic data, indicate a strong preference for complexation with Gal β-(1,3) Gal with the reducing Gal at the primary site, followed by that with Gal α-(1,3) Gal, with the reducing or non-reducing Gal located at the primary binding site. This observation is in consonance with the facility of jacalin to bind mucin type O-glycans containing T-antigen core. © 2016 IUBMB Life, 68(12):971-979, 2016.
Organizational Affiliation:
Molecular Biophysics Unit, Indian Institute of Science, Bangalore, Karnataka, 560012, India.