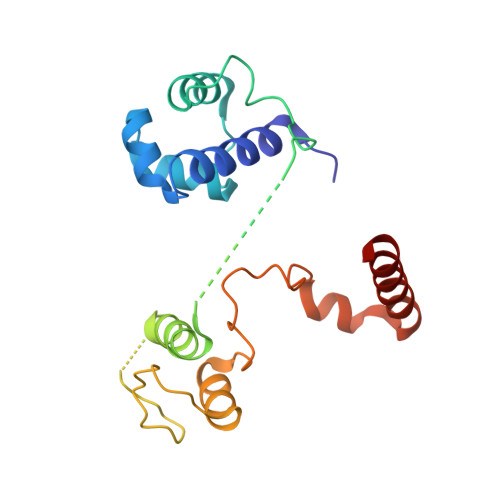
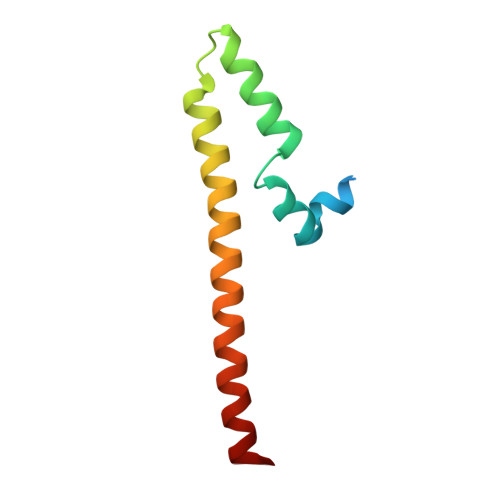
Noncanonical DNA-binding mode of repressor and its disassembly by antirepressor
Kim, M., Kim, H.J., Son, S.H., Yoon, H.J., Lim, Y., Lee, J.W., Seok, Y.-J., Jin, K.S., Yu, Y.G., Kim, S.K., Ryu, S., Lee, H.H.(2016) Proc Natl Acad Sci U S A 113: E2480-E2488
- PubMed: 27099293
- DOI: https://doi.org/10.1073/pnas.1602618113
- Primary Citation of Related Structures:
5D4Z, 5D50 - PubMed Abstract:
DNA-binding repressors are involved in transcriptional repression in many organisms. Disabling a repressor is a crucial step in activating expression of desired genes. Thus, several mechanisms have been identified for the removal of a stably bound repressor (Rep) from the operator. Here, we describe an uncharacterized mechanism of noncanonical DNA binding and induction by a Rep from the temperate Salmonella phage SPC32H; this mechanism was revealed using the crystal structures of homotetrameric Rep (92-198) and a hetero-octameric complex between the Rep and its antirepressor (Ant). The canonical method of inactivating a repressor is through the competitive binding of the antirepressor to the operator-binding site of the repressor; however, these studies revealed several noncanonical features. First, Ant does not compete for the DNA-binding region of Rep. Instead, the tetrameric Ant binds to the C-terminal domains of two asymmetric Rep dimers. Simultaneously, Ant facilitates the binding of the Rep N-terminal domains to Ant, resulting in the release of two Rep dimers from the bound DNA. Second, the dimer pairs of the N-terminal DNA-binding domains originate from different dimers of a Rep tetramer (trans model). This situation is different from that of other canonical Reps, in which two N-terminal DNA-binding domains from the same dimeric unit form a dimer upon DNA binding (cis model). On the basis of these observations, we propose a noncanonical model for the reversible inactivation of a Rep by an Ant.
Organizational Affiliation:
Department of Agricultural Biotechnology, Seoul National University, Seoul 08826, Korea; Center for Food Safety and Toxicology, Seoul National University, Seoul 08826, Korea;