Crystal structure of magnesium-bound Duf89 protein Saccharomyces cerevisiae
Nocek, B., Cuff, M., Cui, H., Xu, X., Savchenko, A., Joachimiak, A., Yakunin, A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein-glutamate O-methyltransferase | 472 | Saccharomyces cerevisiae S288C | Mutation(s): 0 Gene Names: YMR027W, YM9711.17 EC: 2.1.1 | 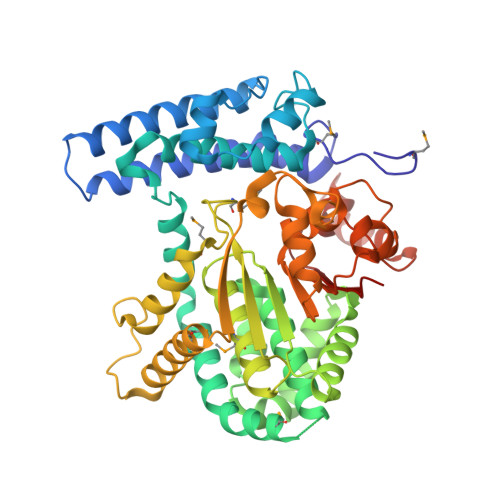 | |
UniProt | |||||
Find proteins for Q04371 (Saccharomyces cerevisiae (strain ATCC 204508 / S288c)) Explore Q04371 Go to UniProtKB: Q04371 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q04371 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MG Query on MG | B [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 54.407 | α = 90 |
| b = 108.582 | β = 97.04 |
| c = 88.234 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| HKL-3000 | phasing |