CRYSTAL STRUCTURE OF VISFATIN IN COMPLEX WITH SAR154782.
Bertrand, T., Marquette, J.P.To be published.
Experimental Data Snapshot
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nicotinamide phosphoribosyltransferase | 493 | Homo sapiens | Mutation(s): 0 Gene Names: NAMPT, PBEF, PBEF1 EC: 2.4.2.12 | 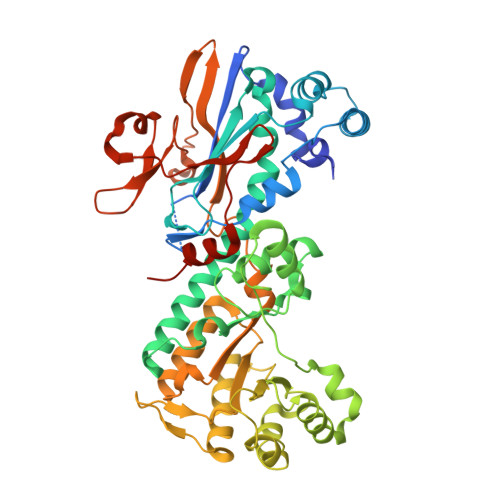 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P43490 (Homo sapiens) Explore P43490 Go to UniProtKB: P43490 | |||||
PHAROS: P43490 GTEx: ENSG00000105835 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P43490 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 7A1 Query on 7A1 | C [auth A], D [auth B] | 6-[4-[(6-azanylpyridin-3-yl)methylcarbamoylamino]-3-fluoranyl-phenyl]-2-(ethylamino)-~{N}-(2-piperidin-1-ylethyl)pyridine-3-carboxamide C28 H35 F N8 O2 JXVZQJNZEWWAKF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 61.51 | α = 90 |
| b = 107.13 | β = 96.69 |
| c = 83.59 | γ = 90 |
| Software Name | Purpose |
|---|---|
| BUSTER-TNT | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| BUSTER-TNT | phasing |