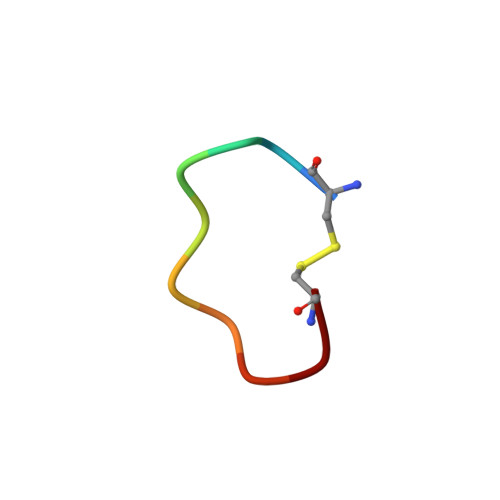
A cyclic peptidic serine protease inhibitor: increasing affinity by increasing peptide flexibility.
Zhao, B., Xu, P., Jiang, L., Paaske, B., Kromann-Hansen, T., Jensen, J.K., Srensen, H.P., Liu, Z., Nielsen, J.T., Christensen, A., Hosseini, M., Srensen, K.K., Nielsen, N.C., Jensen, K.J., Huang, M., Andreasen, P.A.(2014) PLoS One 9: e115872-e115872
- PubMed: 25545505
- DOI: https://doi.org/10.1371/journal.pone.0115872
- Primary Citation of Related Structures:
4X1N, 4X1Q, 4X1R, 4X1S - PubMed Abstract:
Peptides are attracting increasing interest as protease inhibitors. Here, we demonstrate a new inhibitory mechanism and a new type of exosite interactions for a phage-displayed peptide library-derived competitive inhibitor, mupain-1 (CPAYSRYLDC), of the serine protease murine urokinase-type plasminogen activator (uPA). We used X-ray crystal structure analysis, site-directed mutagenesis, liquid state NMR, surface plasmon resonance analysis, and isothermal titration calorimetry and wild type and engineered variants of murine and human uPA. We demonstrate that Arg6 inserts into the S1 specificity pocket, its carbonyl group aligning improperly relative to Ser195 and the oxyanion hole, explaining why the peptide is an inhibitor rather than a substrate. Substitution of the P1 Arg with novel unnatural Arg analogues with aliphatic or aromatic ring structures led to an increased affinity, depending on changes in both P1 - S1 and exosite interactions. Site-directed mutagenesis showed that exosite interactions, while still supporting high affinity binding, differed substantially between different uPA variants. Surprisingly, high affinity binding was facilitated by Ala-substitution of Asp9 of the peptide, in spite of a less favorable binding entropy and loss of a polar interaction. We conclude that increased flexibility of the peptide allows more favorable exosite interactions, which, in combination with the use of novel Arg analogues as P1 residues, can be used to manipulate the affinity and specificity of this peptidic inhibitor, a concept different from conventional attempts at improving inhibitor affinity by reducing the entropic burden.
Organizational Affiliation:
Danish-Chinese Centre for Proteases and Cancer, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, China.