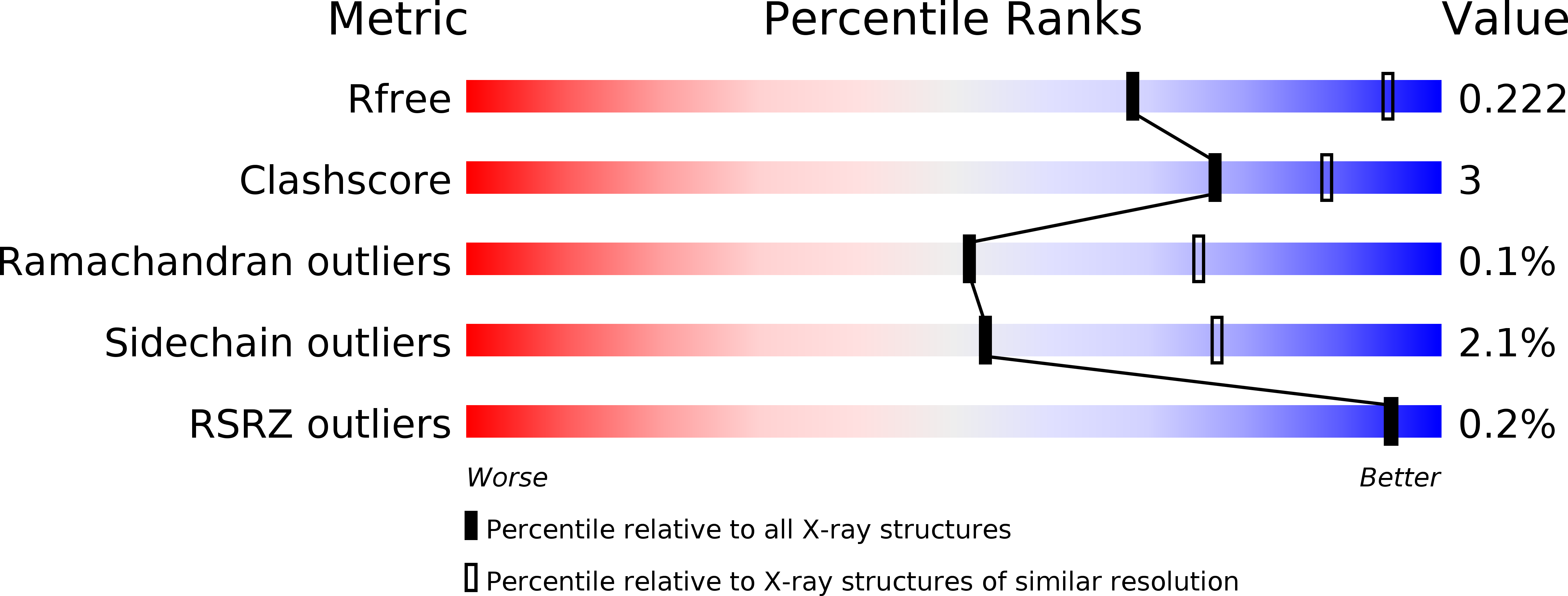
Structures of adenosine kinase from Trypanosoma brucei brucei.
Timm, J., Gonzalez-Pacanowska, D., Wilson, K.S.(2014) Acta Crystallogr Sect F Struct Biol Cryst Commun 70: 34-39
- PubMed: 24419613
- DOI: https://doi.org/10.1107/S2053230X13033621
- Primary Citation of Related Structures:
4N08, 4N09 - PubMed Abstract:
Trypanosoma brucei is a single-cellular parasite of the genus Kinetoplastida and is the causative agent of African sleeping sickness in humans. Adenosine kinase is a key enzyme in the purine-salvage pathway, phosphorylating adenosine to AMP, and also activates cytotoxic analogues such as cordycepin and Ara-A by their phosphorylation. The structures of T. brucei brucei adenosine kinase (TbAK) in its unliganded open conformation and complexed with adenosine and ADP in the closed conformation are both reported to 2.6 Å resolution. The structures give insight into the binding mode of the substrates and the conformational change induced upon substrate binding. This information can be used to guide the improvement of cytotoxic substrate analogues as potential antitrypanosomal drugs.
Organizational Affiliation:
Structural Biology Laboratory, Department of Chemistry, University of York, Heslington, York YO10 5DD, England.