Crystal structure of the complex between human tissue factor extracellular domain and antibody 10H10 FAB fragment
Teplyakov, A., Obmolova, G., Malia, T., Gilliland, G.L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Tissue factor | A [auth T] | 215 | Homo sapiens | Mutation(s): 0 Gene Names: F3 | 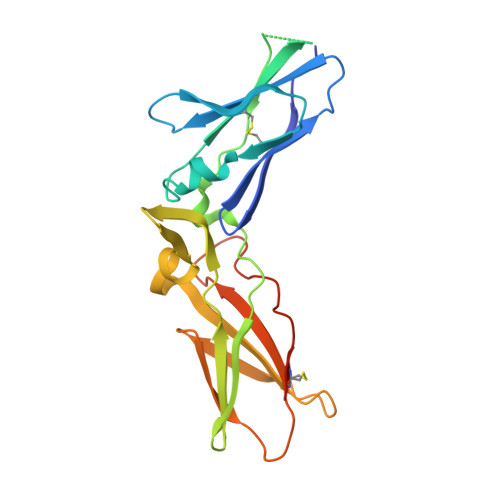 |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P13726 (Homo sapiens) Explore P13726 Go to UniProtKB: P13726 | |||||
PHAROS: P13726 GTEx: ENSG00000117525 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P13726 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 10H10 light chain | B [auth L] | 220 | Mus musculus, Homo sapiens This entity is chimeric | Mutation(s): 0 | 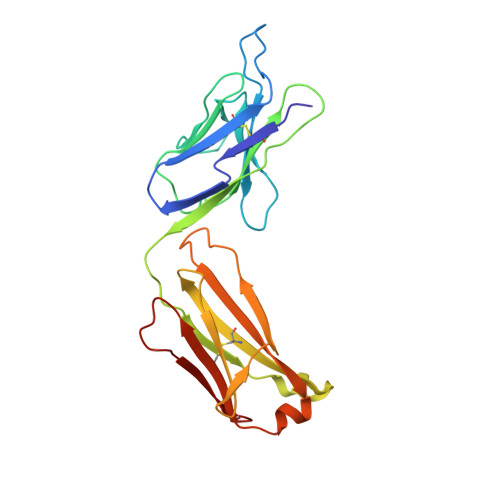 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 10H10 heavy chain | C [auth H] | 229 | Mus musculus, Homo sapiens This entity is chimeric | Mutation(s): 0 | 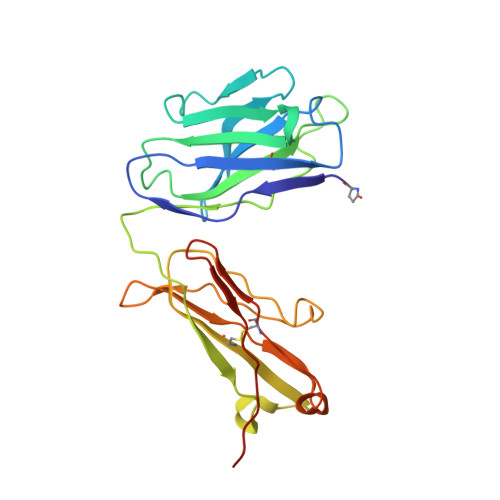 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| PCA Query on PCA | C [auth H] | L-PEPTIDE LINKING | C5 H7 N O3 |  | GLN |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 116.86 | α = 90 |
| b = 116.86 | β = 90 |
| c = 106.86 | γ = 90 |
| Software Name | Purpose |
|---|---|
| StructureStudio | data collection |
| PHASER | phasing |
| REFMAC | refinement |
| XDS | data reduction |
| XDS | data scaling |