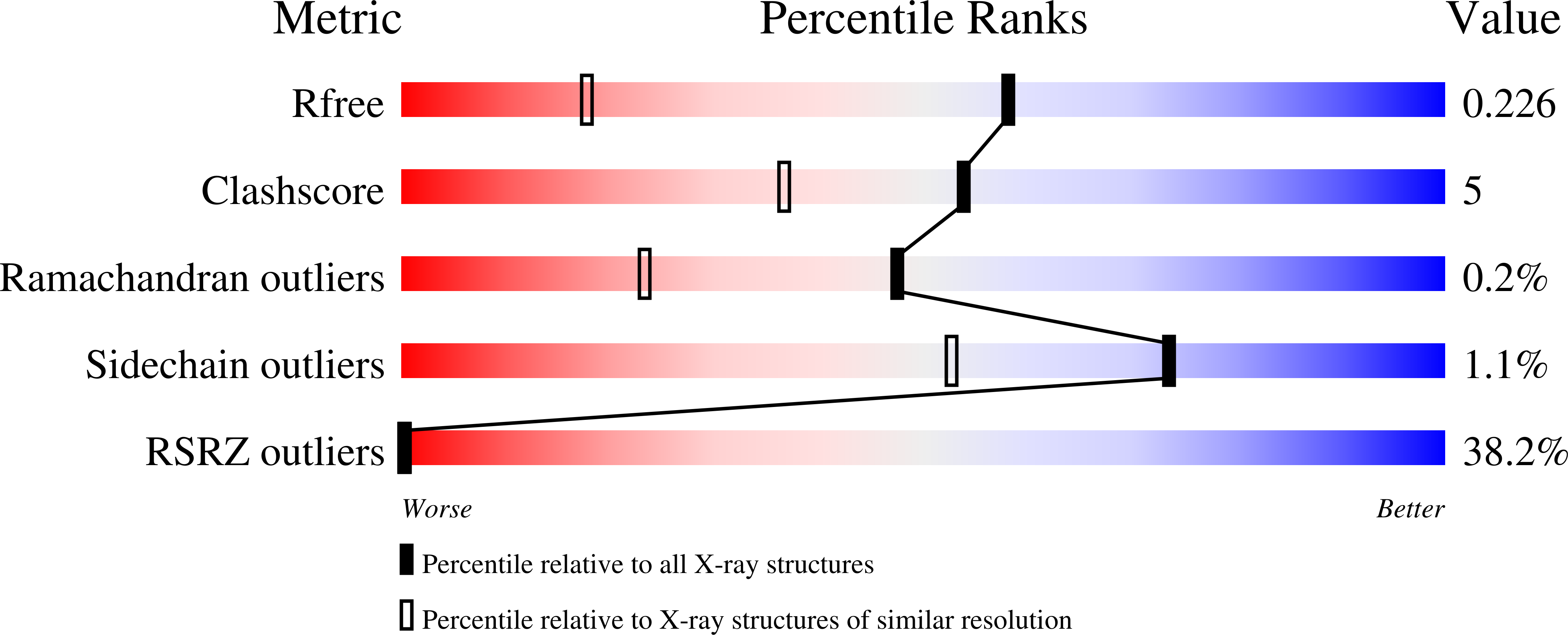
Discovery of a novel class of orally active trypanocidal N-myristoyltransferase inhibitors.
Brand, S., Cleghorn, L.A., McElroy, S.P., Robinson, D.A., Smith, V.C., Hallyburton, I., Harrison, J.R., Norcross, N.R., Spinks, D., Bayliss, T., Norval, S., Stojanovski, L., Torrie, L.S., Frearson, J.A., Brenk, R., Fairlamb, A.H., Ferguson, M.A., Read, K.D., Wyatt, P.G., Gilbert, I.H.(2012) J Med Chem 55: 140-152
- PubMed: 22148754
- DOI: https://doi.org/10.1021/jm201091t
- Primary Citation of Related Structures:
4A2Z, 4A30, 4A31, 4A32, 4A33 - PubMed Abstract:
N-Myristoyltransferase (NMT) represents a promising drug target for human African trypanosomiasis (HAT), which is caused by the parasitic protozoa Trypanosoma brucei. We report the optimization of a high throughput screening hit (1) to give a lead molecule DDD85646 (63), which has potent activity against the enzyme (IC(50) = 2 nM) and T. brucei (EC(50) = 2 nM) in culture. The compound has good oral pharmacokinetics and cures rodent models of peripheral HAT infection. This compound provides an excellent tool for validation of T. brucei NMT as a drug target for HAT as well as a valuable lead for further optimization.
Organizational Affiliation:
Drug Discovery Unit, Division of Biological Chemistry and Drug Discovery, College of Life Sciences, University of Dundee, Sir James Black Centre, Dundee, DD1 5EH, U.K.