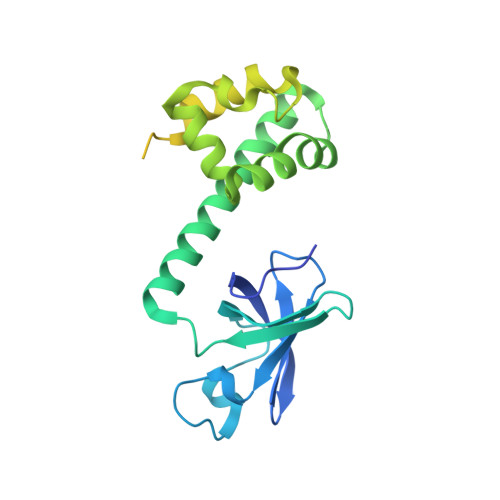
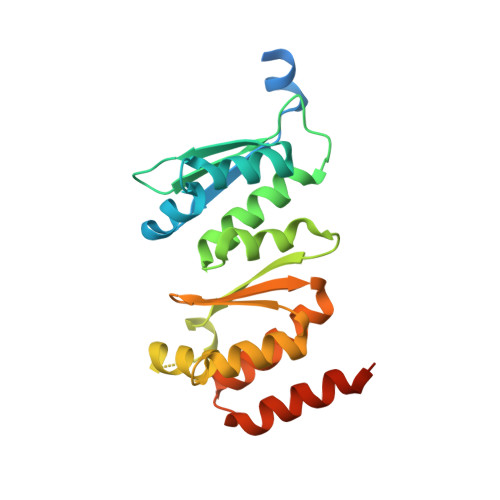
An archaeal Dim2-like protein, aDim2p, forms a ternary complex with a/eIF2 alpha and the 3' end fragment of 16S rRNA
Jia, M.Z., Horita, S., Nagata, K., Tanokura, M.(2010) J Mol Biol 398: 774-785
- PubMed: 20363226
- DOI: https://doi.org/10.1016/j.jmb.2010.03.055
- Primary Citation of Related Structures:
3AEV - PubMed Abstract:
Dim2p is a eukaryal small ribosomal subunit RNA processing factor required for the maturation of 18S rRNA. Here we show that an archaeal homolog of Dim2p, aDim2p, forms a ternary complex with the archaeal homolog of eIF2 alpha, a/eIF2 alpha, and the RNA fragment that possesses the 3' end sequence of 16S rRNA both in solution and in crystal. The 2.8-A crystal structure of the ternary complex reveals that two KH domains of aDim2p, KH-1 and -2, are involved in binding the anti-Shine-Dalgarno core sequence (CCUCC-3') and a highly conserved adjacent sequence (5'-GGAUCA), respectively, of the target rRNA fragment. The surface plasmon resonance results show that the interaction of aDim2p with the target rRNA fragment is very strong, with a dissociation constant of 9.8 x 10(-10) M, and that aDim2p has a strong nucleotide sequence preference for the 3' end sequence of 16S rRNA. On the other hand, aDim2p interacts with the isolated alpha subunit and the intact alpha beta gamma complex of a/eIF2, irrespective of the RNA binding. These results suggest that aDim2p is a possible archaeal pre-rRNA processing factor recognizing the 3' end sequence (5'-GAUCACCUCC-3') of 16S rRNA and may have multiple biological roles in vivo by interacting with other proteins such as a/eIF2 and aRio2p.
Organizational Affiliation:
Department of Applied Biological Chemistry, Graduate School of Agricultural and Life Sciences, University of Tokyo, 1-1-1 Yayoi, Bunkyo-Ku, Tokyo 113-8657, Japan.