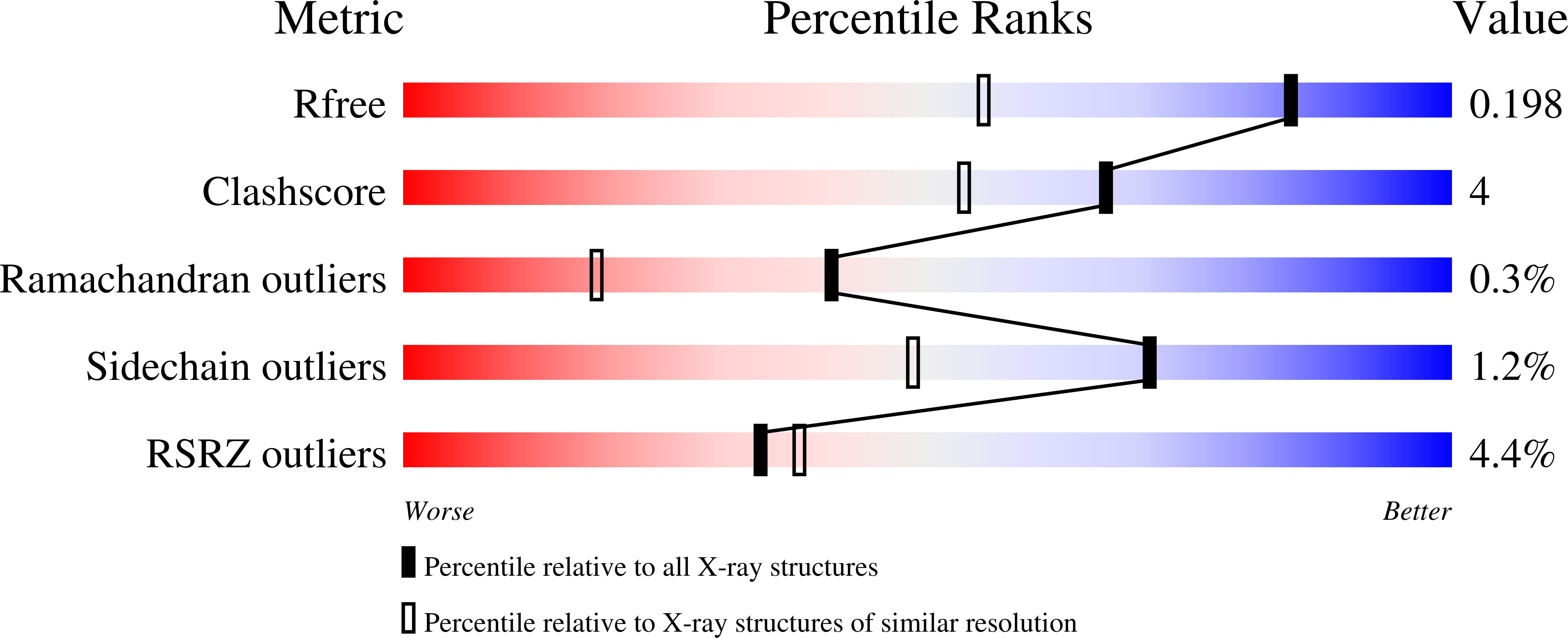
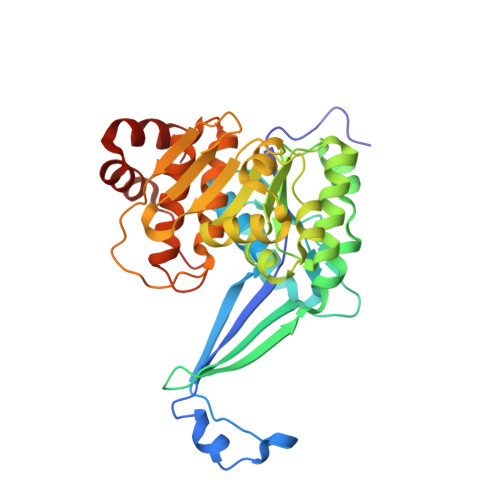
High resolution crystal structures of Mycobacterium tuberculosis adenosine kinase: insights into the mechanism and specificity of this novel prokaryotic enzyme
Reddy, M.C.M., Palaninathan, S.K., Shetty, N.D., Owen, J.L., Watson, M.D., Sacchettini, J.C.(2007) J Biol Chem 282: 27334-27342
- PubMed: 17597075
- DOI: https://doi.org/10.1074/jbc.M703290200
- Primary Citation of Related Structures:
2PKF, 2PKK, 2PKM, 2PKN - PubMed Abstract:
Adenosine kinase (ADK) catalyzes the phosphorylation of adenosine (Ado) to adenosine monophosphate (AMP). It is part of the purine salvage pathway that has been identified only in eukaryotes, with the single exception of Mycobacterium spp. Whereas it is not clear if Mycobacterium tuberculosis (Mtb) ADK is essential, it has been shown that the enzyme can selectively phosphorylate nucleoside analogs to produce products toxic to the cell. We have determined the crystal structure of Mtb ADK unliganded as well as ligand (Ado) bound at 1.5- and 1.9-A resolution, respectively. The structure of the binary complexes with the inhibitor 2-fluoroadenosine (F-Ado) bound and with the adenosine 5'-(beta,gamma-methylene)triphosphate (AMP-PCP) (non-hydrolyzable ATP analog) bound were also solved at 1.9-A resolution. These four structures indicate that Mtb ADK is a dimer formed by an extended beta sheet. The active site of the unliganded ADK is in an open conformation, and upon Ado binding a lid domain of the protein undergoes a large conformation change to close the active site. In the closed conformation, the lid forms direct interactions with the substrate and residues of the active site. Interestingly, AMP-PCP binding alone was not sufficient to produce the closed state of the enzyme. The binding mode of F-Ado was characterized to illustrate the role of additional non-bonding interactions in Mtb ADK compared with human ADK.
Organizational Affiliation:
Department of Biochemistry & Biophysics, Texas A&M University, College Station, Texas 77843.