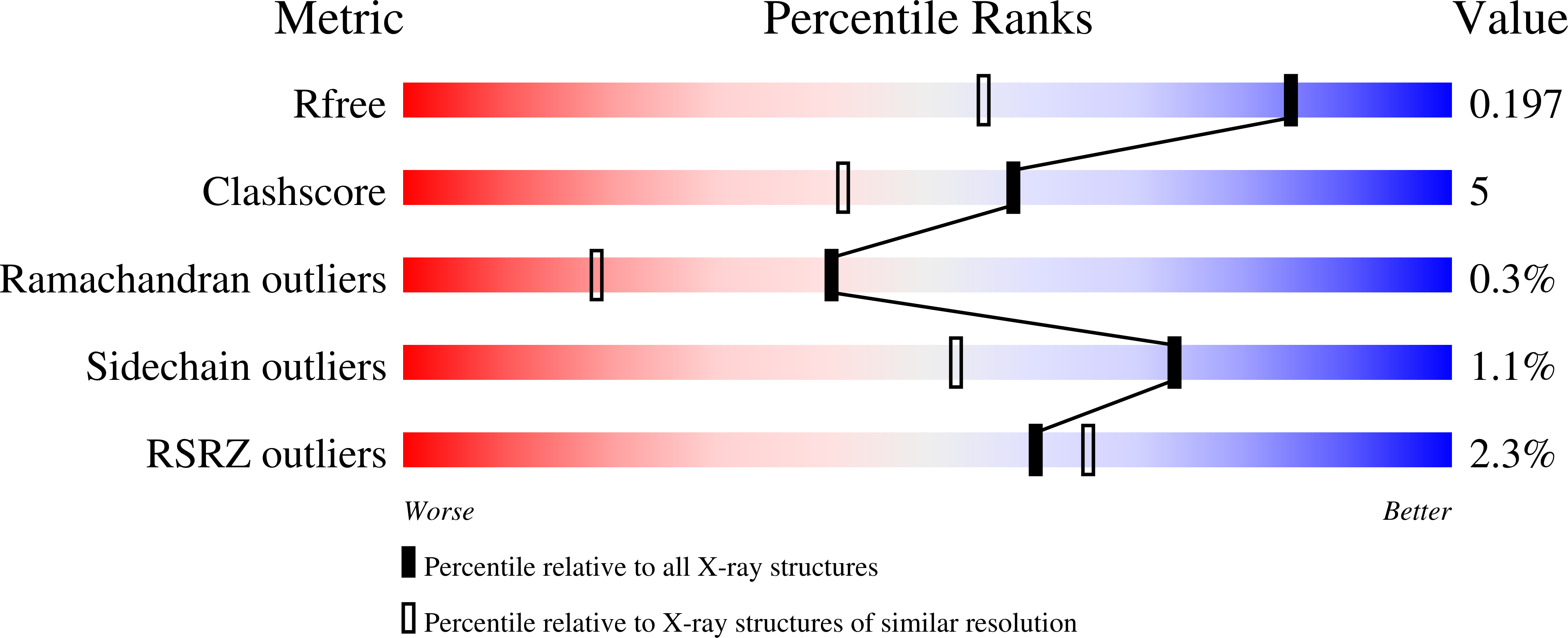
Elucidation of the function of type 1 human methionine aminopeptidase during cell cycle progression.
Hu, X., Addlagatta, A., Lu, J., Matthews, B.W., Liu, J.O.(2006) Proc Natl Acad Sci U S A 103: 18148-18153
- PubMed: 17114291
- DOI: https://doi.org/10.1073/pnas.0608389103
- Primary Citation of Related Structures:
2NQ6, 2NQ7 - PubMed Abstract:
Processing of the N-terminal initiator methionine is an essential cellular process conserved from prokaryotes to eukaryotes. The enzymes that remove N-terminal methionine are known as methionine aminopeptidases (MetAPs). Human MetAP2 has been shown to be required for the proliferation of endothelial cells and angiogenesis. The physiological function of MetAP1, however, has remained elusive. In this report we demonstrate that a family of inhibitors with a core structure of pyridine-2-carboxylic acid previously developed for the bacterial and yeast MetAP1 is also specific for human MetAP1 (HsMetAP1), as confirmed by both enzymatic assay and high-resolution x-ray crystallography. Treatment of tumor cell lines with the MetAP1-specific inhibitors led to an accumulation of cells in the G(2)/M phase, suggesting that HsMetAP1 may play an important role in G(2)/M phase transition. Overexpression of HsMetAP1, but not HsMetAP2, conferred resistance of cells to the inhibitors, and the inhibitors caused retention of N-terminal methionine of a known MetAP substrate, suggesting that HsMetAP1 is the cellular target for the inhibitors. In addition, when HsMetAP1 was knocked down by gene-specific siRNA, cells exhibited slower progression during G(2)/M phase, a phenotype similar to cells treated with MetAP1 inhibitors. Importantly, MetAP1 inhibitors were able to induce apoptosis of leukemia cell lines, presumably as a consequence of their interference with the G(2)/M phase checkpoint. Together, these results suggest that MetAP1 plays an important role in G(2)/M phase of the cell cycle and that it may serve as a promising target for the discovery and development of new anticancer agents.
Organizational Affiliation:
Department of Pharmacology and Molecular Sciences, Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA.